QUESTION IMAGE
Question
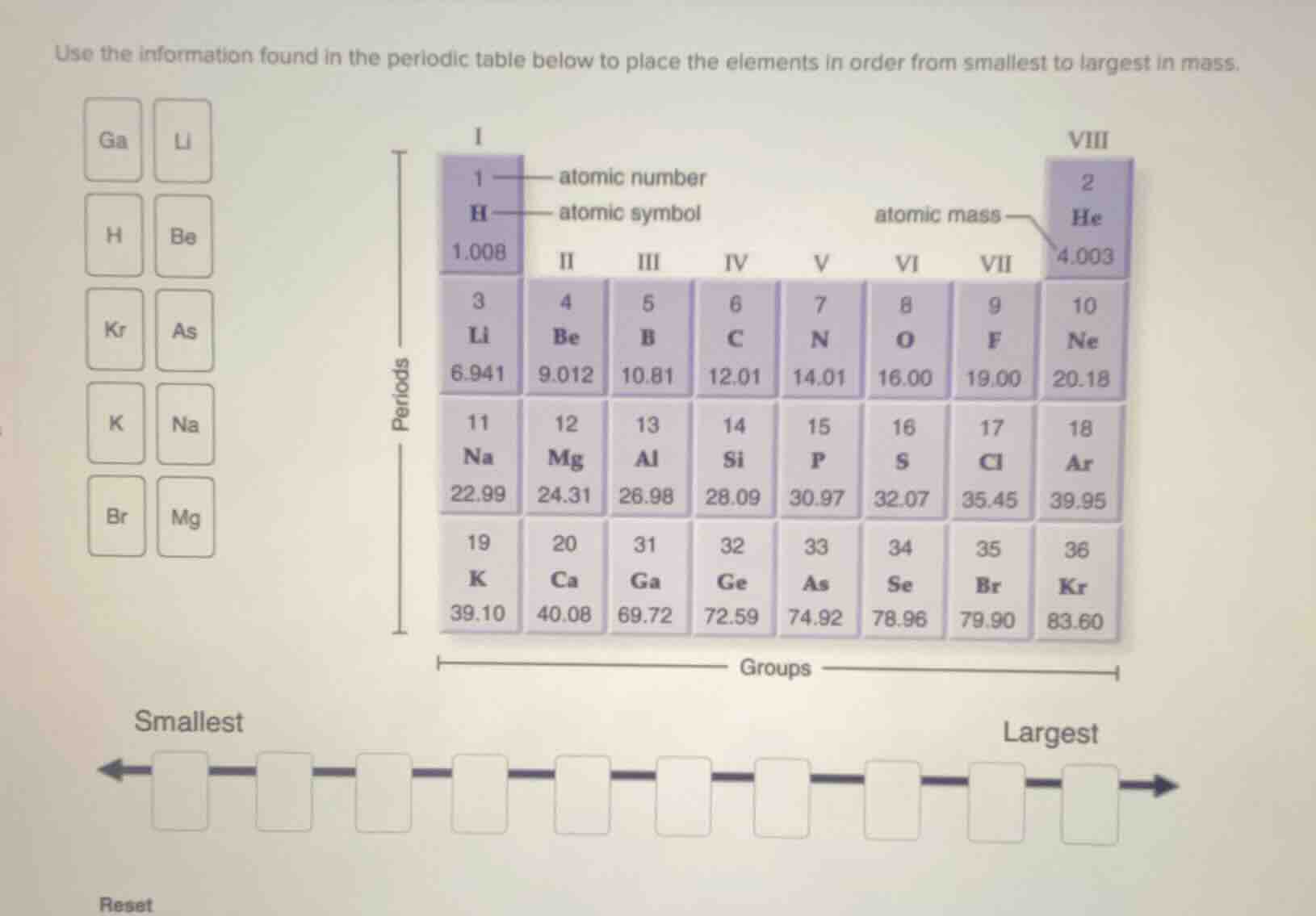
use the information found in the periodic table below to place the elements in order from smallest to largest in mass.
ga li
h be
kr as
k na
br mg
smallest
largest
reset
Step1: Find atomic masses
From the periodic table:
- \( H \): \( 1.008 \)
- \( Li \): \( 6.941 \)
- \( Be \): \( 9.012 \)
- \( Na \): \( 22.99 \)
- \( Mg \): \( 24.31 \)
- \( As \): \( 74.92 \)
- \( Ga \): \( 69.72 \)
- \( K \): \( 39.10 \)
- \( Br \): \( 79.90 \)
- \( Kr \): \( 83.60 \)
Step2: Order by mass (smallest to largest)
Sort the masses:
\( 1.008 (H) < 6.941 (Li) < 9.012 (Be) < 22.99 (Na) < 24.31 (Mg) < 39.10 (K) < 69.72 (Ga) < 74.92 (As) < 79.90 (Br) < 83.60 (Kr) \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Smallest to Largest: \( H \), \( Li \), \( Be \), \( Na \), \( Mg \), \( K \), \( Ga \), \( As \), \( Br \), \( Kr \)