QUESTION IMAGE
Question
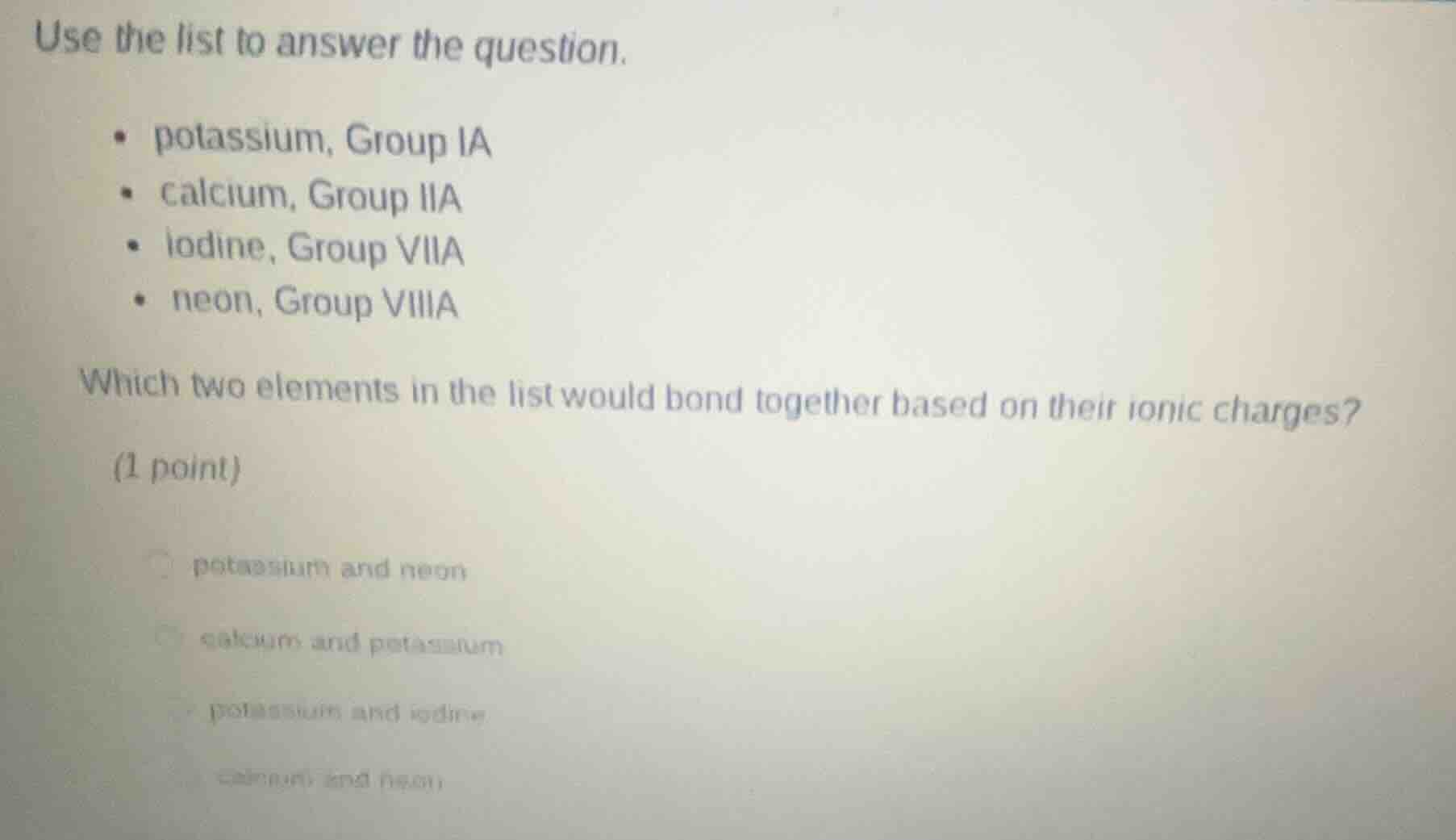
use the list to answer the question.
- potassium, group ia
- calcium, group iia
- iodine, group viia
- neon, group viiia
which two elements in the list would bond together based on their ionic charges?
(1 point)
- potassium and neon
- calcium and potassium
- potassium and iodine
- calcium and neon
Brief Explanations
Ionic bonds form between metals (that lose electrons to form positive ions) and nonmetals (that gain electrons to form negative ions). Noble gases (Group VIIIA) are inert and do not form ionic bonds.
- Potassium (Group IA) is a metal that forms a +1 ion ($\text{K}^+$).
- Iodine (Group VIIA) is a nonmetal that forms a -1 ion ($\text{I}^-$).
- Their charges cancel out, allowing them to form an ionic bond.
Other pairs: potassium/neon (neon is inert), calcium/potassium (both positive metal ions, no ionic bond), calcium/neon (neon is inert) cannot form ionic bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
potassium and iodine