QUESTION IMAGE
Question
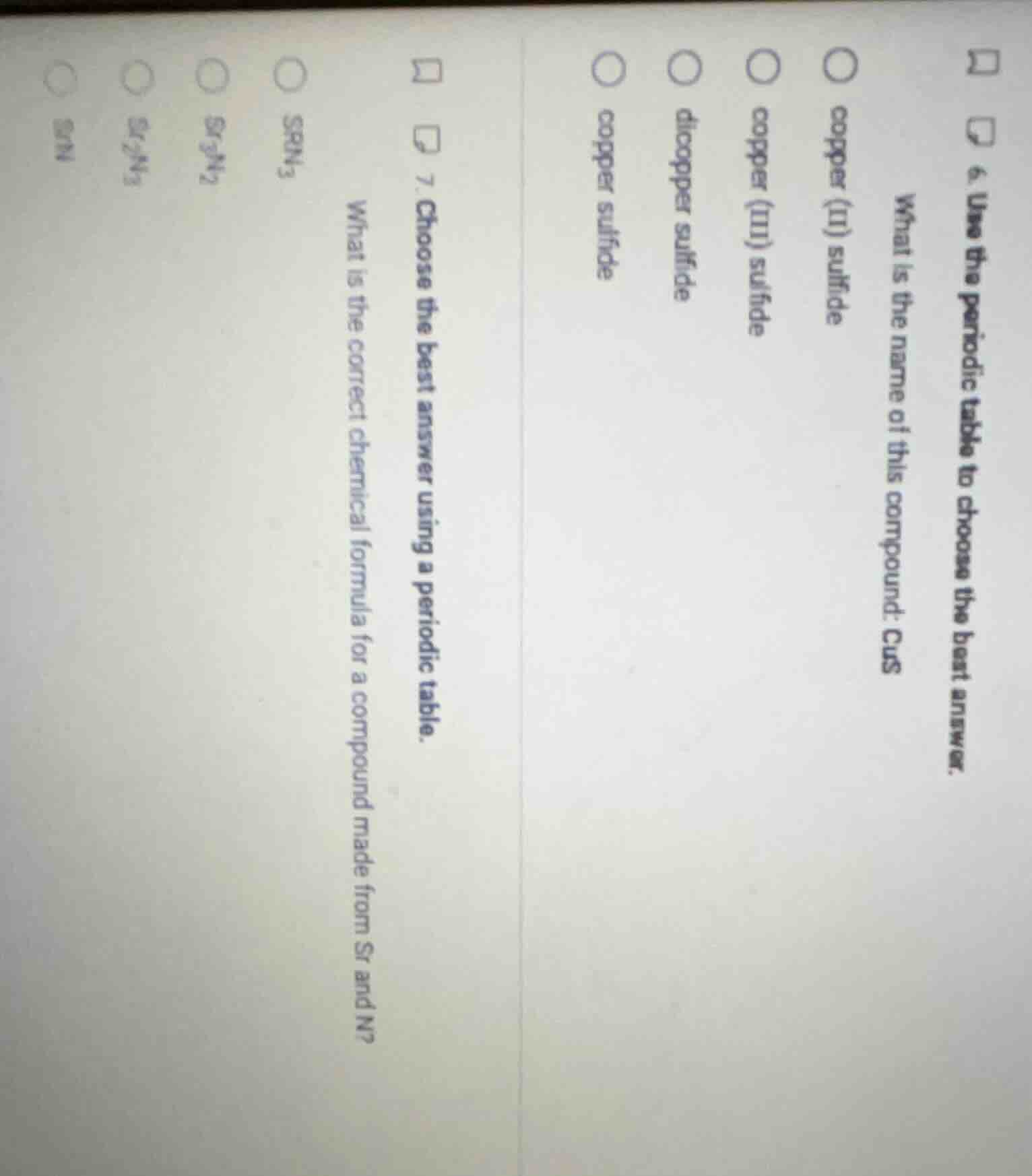
- use the periodic table to choose the best answer. what is the name of this compound: cus copper (ii) sulfide copper (iii) sulfide dicopper sulfide copper sulfide 7. choose the best answer using a periodic table. what is the correct chemical formula for a compound made from sr and n? srn₃ sr₃n₂ sr₂n₃ srn
Step1: Analyze CuS for Q6
Copper forms ions with +1 or +2 charges; sulfide has a -2 charge ($\text{S}^{2-}$). For charge neutrality, $\text{Cu}^{2+}$ pairs with $\text{S}^{2-}$, so copper has a +2 (II) oxidation state.
Step2: Analyze Sr and N for Q7
Strontium (Sr) is group 2, forms $\text{Sr}^{2+}$. Nitrogen (N) is group 15, forms $\text{N}^{3-}$. Cross charges to balance: $3\times(+2) + 2\times(-3)=0$, giving the formula $\text{Sr}_3\text{N}_2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- copper (II) sulfide
- $\text{Sr}_3\text{N}_2$