QUESTION IMAGE
Question
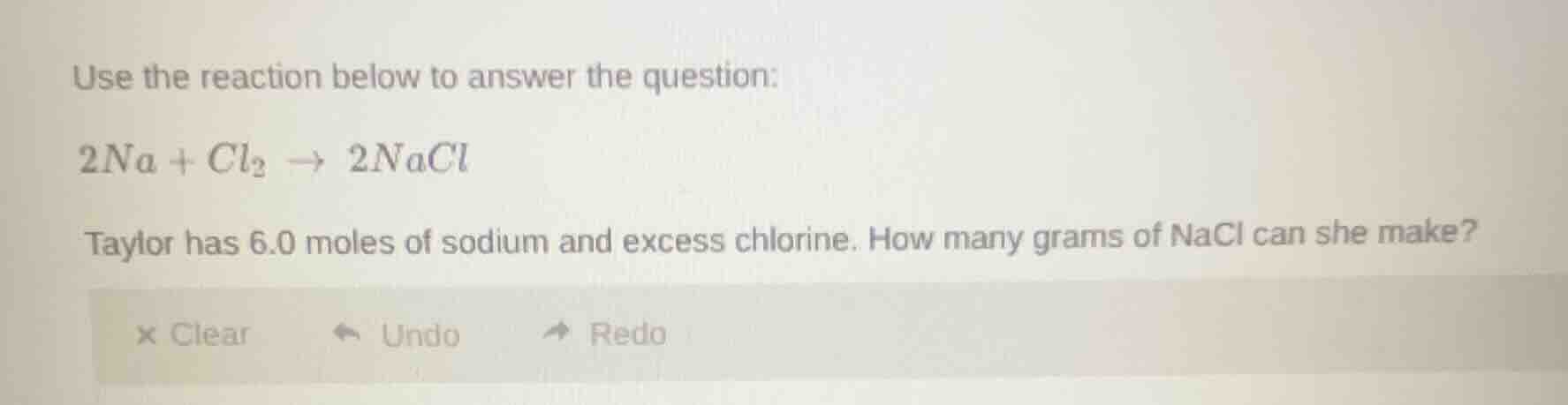
use the reaction below to answer the question:
$2na + cl_2 \
ightarrow 2nacl$
taylor has 6.0 moles of sodium and excess chlorine. how many grams of nacl can she make?
Step1: Mole ratio of Na to NaCl
From reaction $2Na + Cl_2
ightarrow 2NaCl$, mole ratio $\frac{n_{NaCl}}{n_{Na}} = \frac{2}{2} = 1$
Step2: Calculate moles of NaCl
$n_{NaCl} = n_{Na} = 6.0\ \text{mol}$
Step3: Molar mass of NaCl
$M_{NaCl} = 23.0\ \text{g/mol} + 35.5\ \text{g/mol} = 58.5\ \text{g/mol}$
Step4: Calculate mass of NaCl
$m_{NaCl} = n_{NaCl} \times M_{NaCl} = 6.0\ \text{mol} \times 58.5\ \text{g/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
351 grams