QUESTION IMAGE
Question
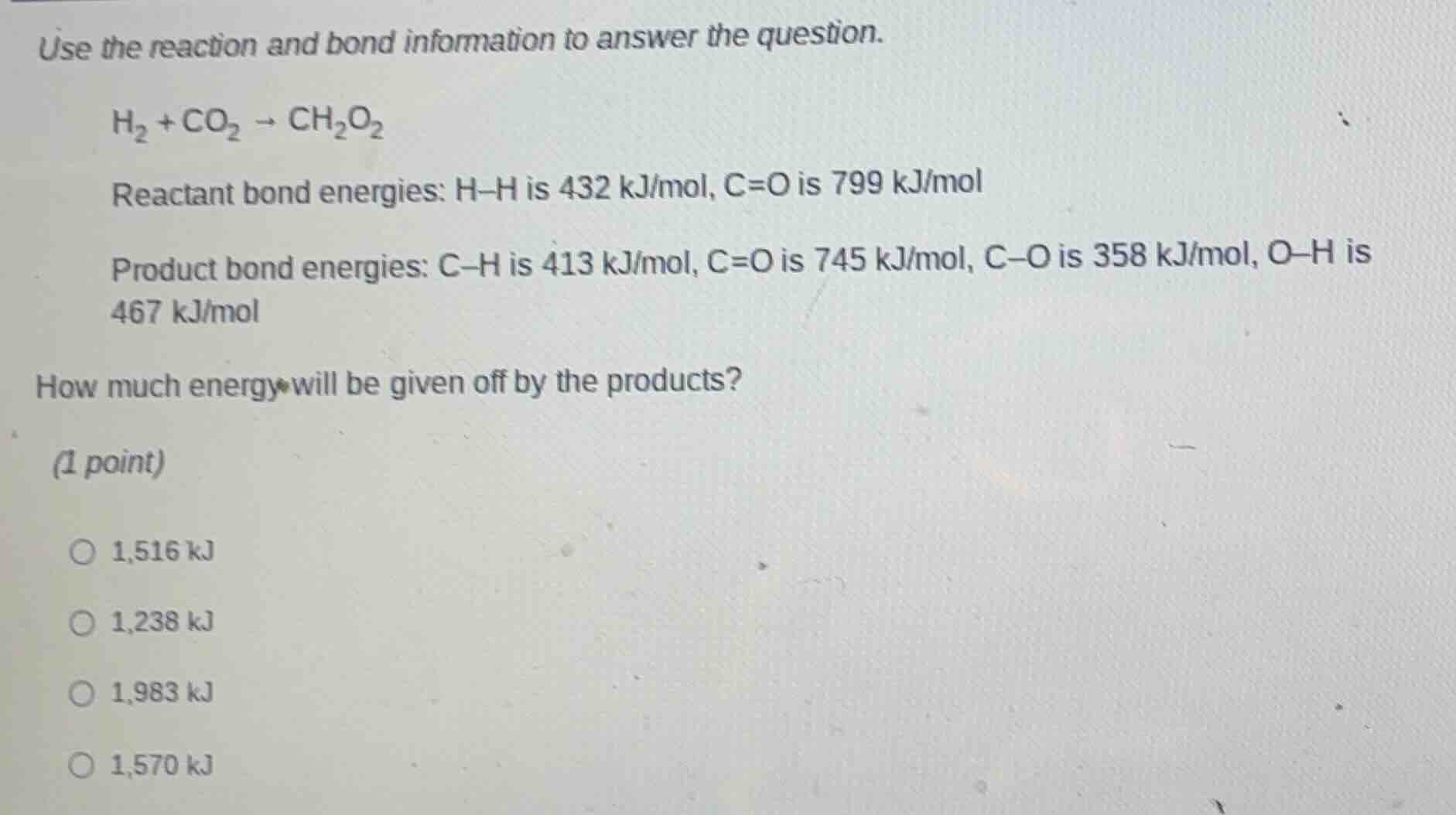
use the reaction and bond information to answer the question.
\\(\ce{h_{2} + co_{2} \
ightarrow ch_{2}o_{2}}\\)
reactant bond energies: \\(\ce{h-h}\\) is 432 kj/mol, \\(\ce{c=o}\\) is 799 kj/mol
product bond energies: \\(\ce{c-h}\\) is 413 kj/mol, \\(\ce{c=o}\\) is 745 kj/mol, \\(\ce{c-o}\\) is 358 kj/mol, \\(\ce{o-h}\\) is 467 kj/mol
how much energy will be given off by the products?
(1 point)
\\(\circ\\) 1,516 kj
\\(\circ\\) 1,238 kj
\\(\circ\\) 1,983 kj
\\(\circ\\) 1,570 kj
Step1: Determine bonds in products
In \( \text{CH}_2\text{O}_2 \), let's identify the bonds. The structure can be considered as having 2 \( \text{C-H} \) bonds, 1 \( \text{C=O} \) bond, and 1 \( \text{O-H} \) bond? Wait, no, let's re - examine. Wait, the correct structure of \( \text{CH}_2\text{O}_2 \) (formic acid) has \( \text{C-H} \), \( \text{C=O} \), \( \text{C-O} \), and \( \text{O-H} \)? Wait, no, let's count the bonds properly. Wait, the formula is \( \text{CH}_2\text{O}_2 \), so the bonds: 2 \( \text{C-H} \), 1 \( \text{C=O} \), 1 \( \text{C-O} \), and 1 \( \text{O-H} \)? Wait, no, let's use the bond energies given. Wait, the product bond energies are \( \text{C-H} = 413 \) kJ/mol, \( \text{C=O}=745 \) kJ/mol, \( \text{C-O}=358 \) kJ/mol, \( \text{O-H}=467 \) kJ/mol.
Wait, let's write the product \( \text{CH}_2\text{O}_2 \) structure. The correct bonding for formic acid (\( \text{HCOOH} \)) is \( \text{H - C(=O)-O - H} \). So the bonds are: 1 \( \text{C-H} \), 1 \( \text{C=O} \), 1 \( \text{C-O} \), and 1 \( \text{O-H} \)? Wait, no, the formula is \( \text{CH}_2\text{O}_2 \), so there are 2 H atoms. So \( \text{H - C(=O)-O - H} \): so bonds are \( \text{C-H} \) (1), \( \text{C=O} \) (1), \( \text{C-O} \) (1), \( \text{O-H} \) (1), and wait, no, the first H is \( \text{C-H} \), the second H is \( \text{O-H} \). Wait, maybe I made a mistake. Let's count the number of each bond:
For \( \text{CH}_2\text{O}_2 \):
- Number of \( \text{C-H} \) bonds: 2
- Number of \( \text{C=O} \) bonds: 1
- Number of \( \text{C-O} \) bonds: 1
- Number of \( \text{O-H} \) bonds: 1
Wait, no, let's calculate the total energy released when forming the products (which is the sum of the bond energies of the products, since energy is released when bonds are formed).
So, the energy released when forming the products is the sum of the bond energies of all the bonds in the product, multiplied by the number of each bond.
Number of \( \text{C-H} \) bonds: 2, so energy for \( \text{C-H} \) bonds: \( 2\times413 \) kJ/mol
Number of \( \text{C=O} \) bonds: 1, energy for \( \text{C=O} \) bond: \( 1\times745 \) kJ/mol
Number of \( \text{C-O} \) bonds: 1, energy for \( \text{C-O} \) bond: \( 1\times358 \) kJ/mol
Number of \( \text{O-H} \) bonds: 1, energy for \( \text{O-H} \) bond: \( 1\times467 \) kJ/mol
Wait, no, wait, maybe the structure is different. Wait, let's re - check the reaction: \( \text{H}_2+\text{CO}_2
ightarrow\text{CH}_2\text{O}_2 \)
\( \text{CO}_2 \) has 2 \( \text{C=O} \) bonds. \( \text{H}_2 \) has 1 \( \text{H-H} \) bond.
For the product \( \text{CH}_2\text{O}_2 \), let's count the bonds correctly. Let's use the following approach: the energy given off by the products is the total bond energy of the products (since forming bonds releases energy).
So, let's list the bonds in \( \text{CH}_2\text{O}_2 \):
- \( \text{C-H} \): 2 bonds (because there are 2 H atoms bonded to C? Wait, no, in \( \text{CH}_2\text{O}_2 \), the formula is \( \text{CH}_2\text{O}_2 \), so the number of \( \text{C-H} \) bonds is 2? Wait, no, the correct structure of formic acid is \( \text{HCOOH} \), which is \( \text{H - C(=O)-O - H} \), so there is 1 \( \text{C-H} \) bond, 1 \( \text{C=O} \) bond, 1 \( \text{C-O} \) bond, and 1 \( \text{O-H} \) bond. Wait, this is a mistake. Let's calculate the number of each bond:
From the formula \( \text{CH}_2\text{O}_2 \), the number of atoms: C = 1, H = 2, O = 2.
Bonds:
- \( \text{C-H} \): 2 (since there are 2 H atoms attached to C? No, in formic acid, one H is attached to C, and one H is attached to O. So \( \text{C-…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1983 kJ