QUESTION IMAGE
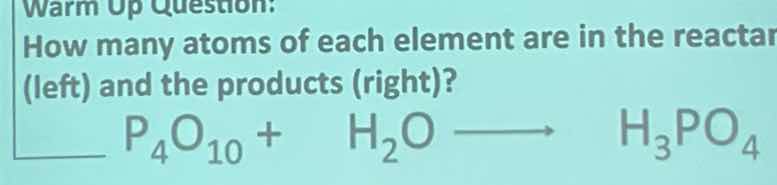
Question
warm up question:
how many atoms of each element are in the reactant (left) and the products (right)?
$\boldsymbol{___ p_4o_{10} + h_2o longrightarrow h_3po_4}$
Step1: Balance P atoms first
Count P: Left has 4, right has 1. Multiply $\text{H}_3\text{PO}_4$ by 4:
$$\text{P}_4\text{O}_{10} + \text{H}_2\text{O} \longrightarrow 4\text{H}_3\text{PO}_4$$
Step2: Balance H atoms
Count H: Right has $4\times3=12$, left has 2. Multiply $\text{H}_2\text{O}$ by 6:
$$\text{P}_4\text{O}_{10} + 6\text{H}_2\text{O} \longrightarrow 4\text{H}_3\text{PO}_4$$
Step3: Verify O atoms
Left: $10 + 6\times1=16$; Right: $4\times4=16$. Balanced.
Step4: Count reactant atoms
P: 4, O: $10+6=16$, H: $6\times2=12$
Step5: Count product atoms
P: $4\times1=4$, O: $4\times4=16$, H: $4\times3=12$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Reactants (left side):
- Phosphorus (P): 4 atoms
- Oxygen (O): 16 atoms
- Hydrogen (H): 12 atoms
Products (right side):
- Phosphorus (P): 4 atoms
- Oxygen (O): 16 atoms
- Hydrogen (H): 12 atoms