QUESTION IMAGE
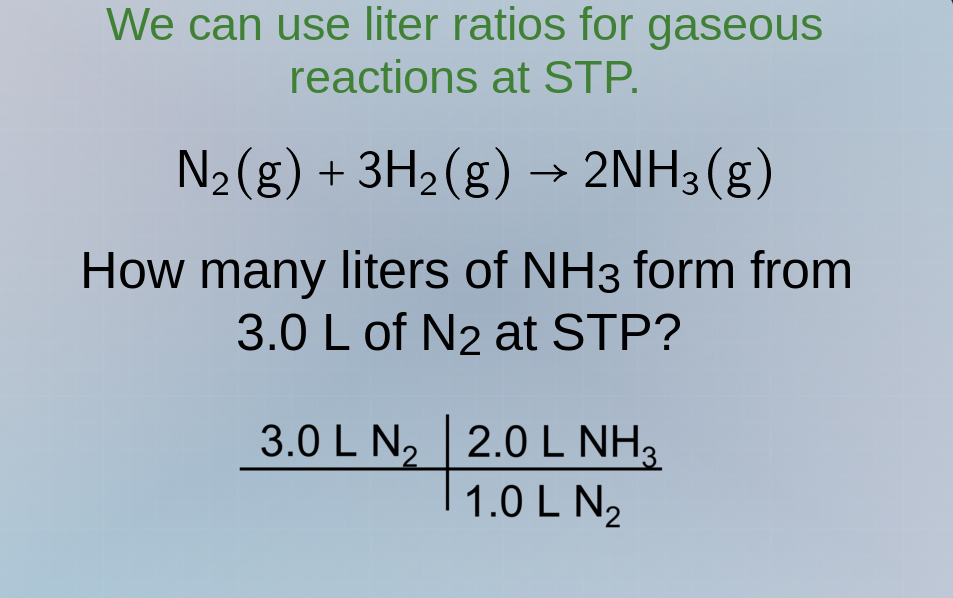
Question
we can use liter ratios for gaseous reactions at stp.
n₂(g) + 3h₂(g) → 2nh₃(g)
how many liters of nh₃ form from 3.0 l of n₂ at stp?
(3.0 l n₂) * (2.0 l nh₃ / 1.0 l n₂)
Step1: Identify the mole ratio (or volume ratio for gases at STP) from the balanced equation.
From the reaction \( \text{N}_2(\text{g}) + 3\text{H}_2(\text{g})
ightarrow 2\text{NH}_3(\text{g}) \), the volume ratio of \( \text{N}_2 \) to \( \text{NH}_3 \) is \( 1:2 \) (since at STP, volume ratios are equal to mole ratios for gases).
Step2: Use the given volume of \( \text{N}_2 \) and the volume ratio to calculate the volume of \( \text{NH}_3 \).
We have \( 3.0 \, \text{L} \) of \( \text{N}_2 \), and the ratio of \( \text{NH}_3 \) to \( \text{N}_2 \) is \( \frac{2.0 \, \text{L} \, \text{NH}_3}{1.0 \, \text{L} \, \text{N}_2} \). So we multiply the volume of \( \text{N}_2 \) by this ratio:
\( 3.0 \, \text{L} \, \text{N}_2 \times \frac{2.0 \, \text{L} \, \text{NH}_3}{1.0 \, \text{L} \, \text{N}_2} \)
Step3: Perform the calculation.
\( 3.0 \times 2.0 = 6.0 \) (the units of \( \text{L} \, \text{N}_2 \) cancel out, leaving \( \text{L} \, \text{NH}_3 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\( 6.0 \, \text{L} \)