QUESTION IMAGE
Question
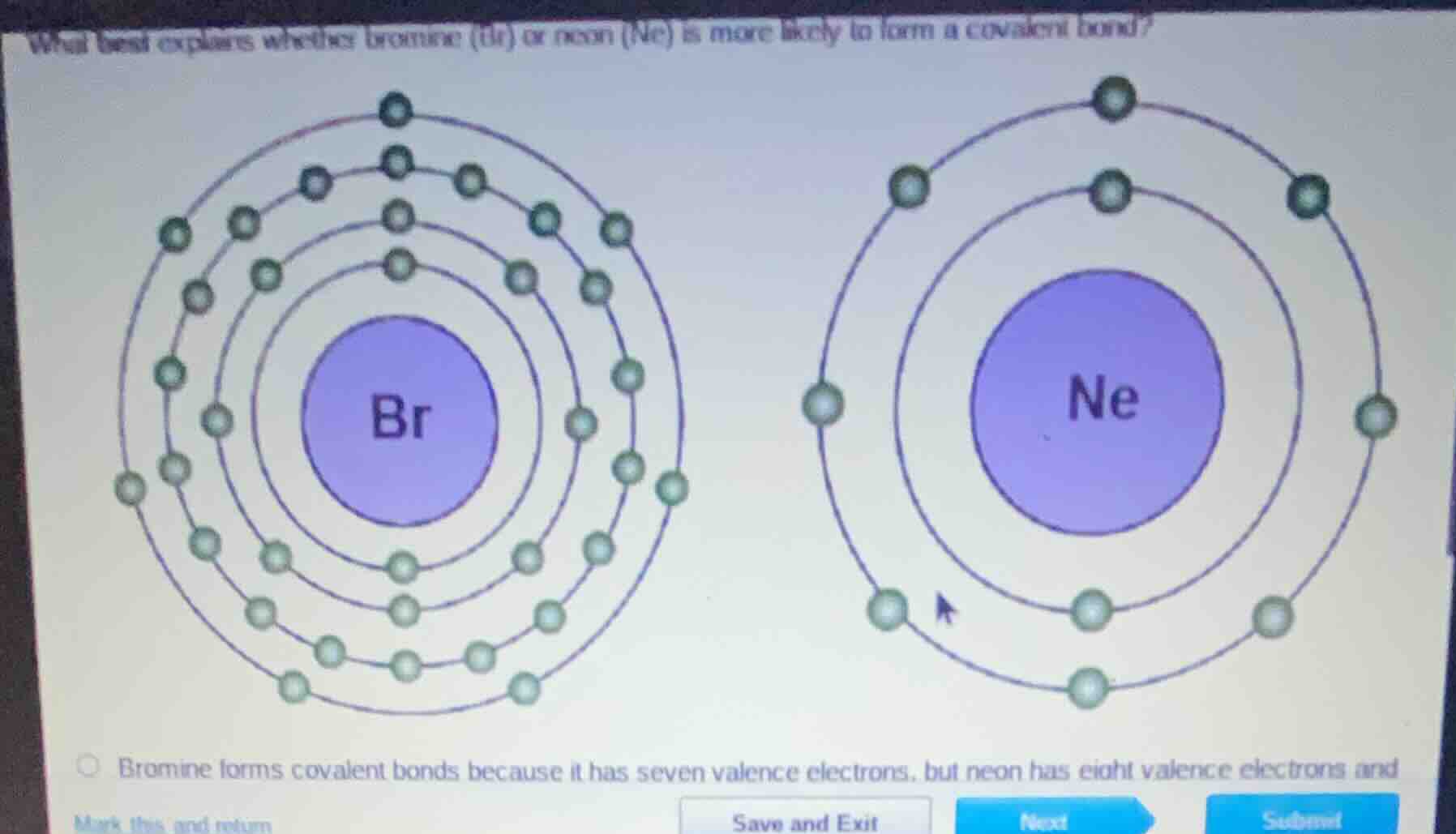
what best explains whether bromine (br) or neon (ne) is more likely to form a covalent bond? bromine forms covalent bonds because it has seven valence electrons, but neon has eight valence electrons and
Covalent bonds form when atoms share electrons to achieve a full valence shell (8 electrons, octet rule). Bromine (Br) has 7 valence electrons, so it needs 1 more electron to complete its octet, making it likely to share electrons in a covalent bond. Neon (Ne) is a noble gas with a full 8-valence-electron shell, so it is stable and does not form covalent bonds.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Bromine forms covalent bonds because it has seven valence electrons, but neon has eight valence electrons and is stable, so it does not form covalent bonds.