QUESTION IMAGE
Question
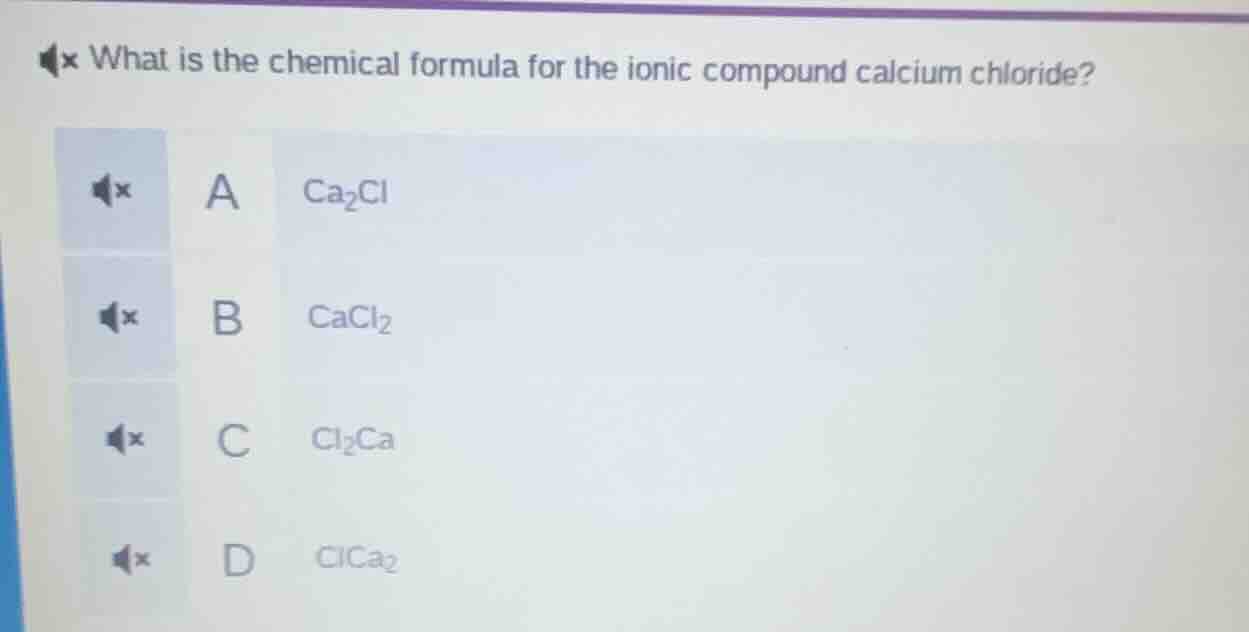
what is the chemical formula for the ionic compound calcium chloride? a ca₂cl b cacl₂ c cl₂ca d clca₂
Step1: Identify ion charges
Calcium (Ca) forms $\text{Ca}^{2+}$; chloride (Cl) forms $\text{Cl}^-$
Step2: Balance charge neutrality
Need 2 $\text{Cl}^-$ to balance 1 $\text{Ca}^{2+}$ (total charge: $+2 + 2(-1) = 0$)
Step3: Write formula (cation first)
Cation is written first, followed by anion with subscript for count.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. $\text{CaCl}_2$