QUESTION IMAGE
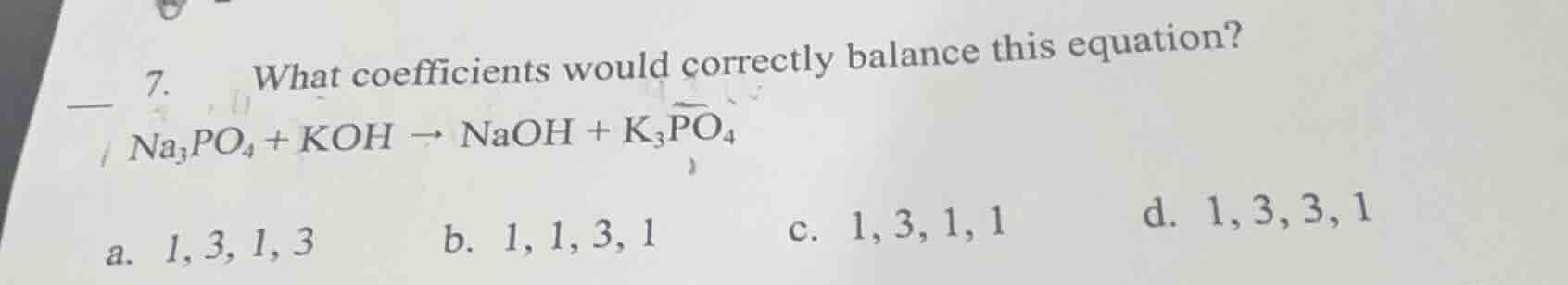
Question
- what coefficients would correctly balance this equation? \\( \ce{na_{3}po_{4} + koh \
ightarrow naoh + k_{3}po_{4}} \\) \\( \text{a. } 1, 3, 1, 3 \\) \\( \text{b. } 1, 1, 3, 1 \\) \\( \text{c. } 1, 3, 1, 1 \\) \\( \text{d. } 1, 3, 3, 1 \\)
Step1: Balance Na atoms
Count Na on left: 3 (from $\text{Na}_3\text{PO}_4$). So $\text{NaOH}$ needs coefficient 3:
$\text{Na}_3\text{PO}_4 + \text{KOH}
ightarrow 3\text{NaOH} + \text{K}_3\text{PO}_4$
Step2: Balance OH⁻/K atoms
Right has 3 OH⁻ (from 3$\text{NaOH}$), so $\text{KOH}$ needs coefficient 3. This gives 3 K on left, matching 3 K in $\text{K}_3\text{PO}_4$ on right:
$\text{Na}_3\text{PO}_4 + 3\text{KOH}
ightarrow 3\text{NaOH} + \text{K}_3\text{PO}_4$
Step3: Verify all atoms
Check $\text{PO}_4^{3-}$: 1 on each side. All atoms are balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. 1, 3, 3, 1