QUESTION IMAGE
Question
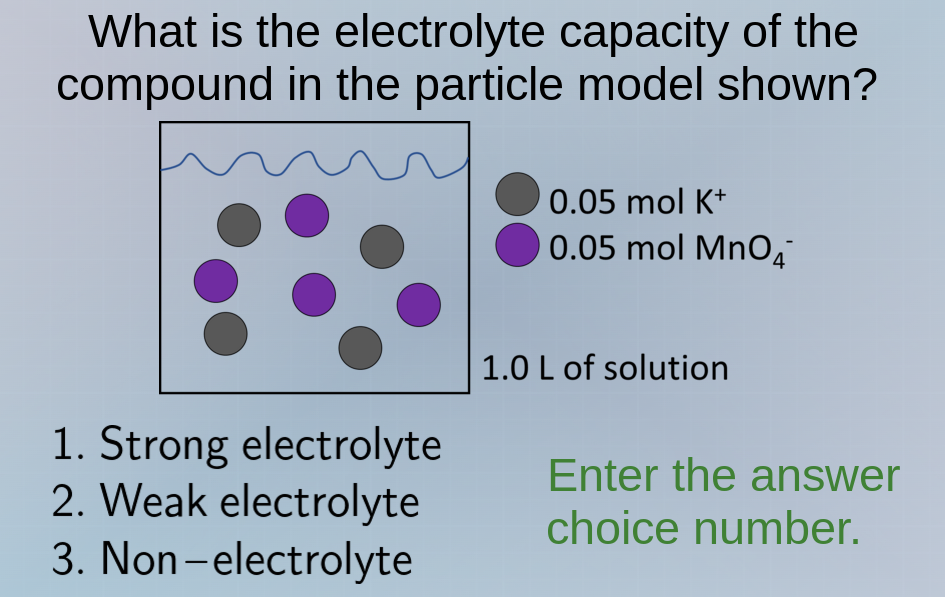
what is the electrolyte capacity of the compound in the particle model shown? 1. strong electrolyte 2. weak electrolyte 3. non–electrolyte enter the answer choice number. 0.05 mol k⁺ 0.05 mol mno₄⁻ 1.0 l of solution
To determine the electrolyte type, we analyze the particle model. The compound here is likely \( KMnO_4 \) (since \( K^+ \) and \( MnO_4^- \) are present). \( KMnO_4 \) is a strong electrolyte because it dissociates completely into ions (\( K^+ \) and \( MnO_4^- \)) in solution, as indicated by the presence of separate ions (no undissociated molecules, just ions) in the model. Strong electrolytes dissociate completely, weak electrolytes partially, and non - electrolytes do not dissociate into ions. So this compound is a strong electrolyte.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1