QUESTION IMAGE
Question
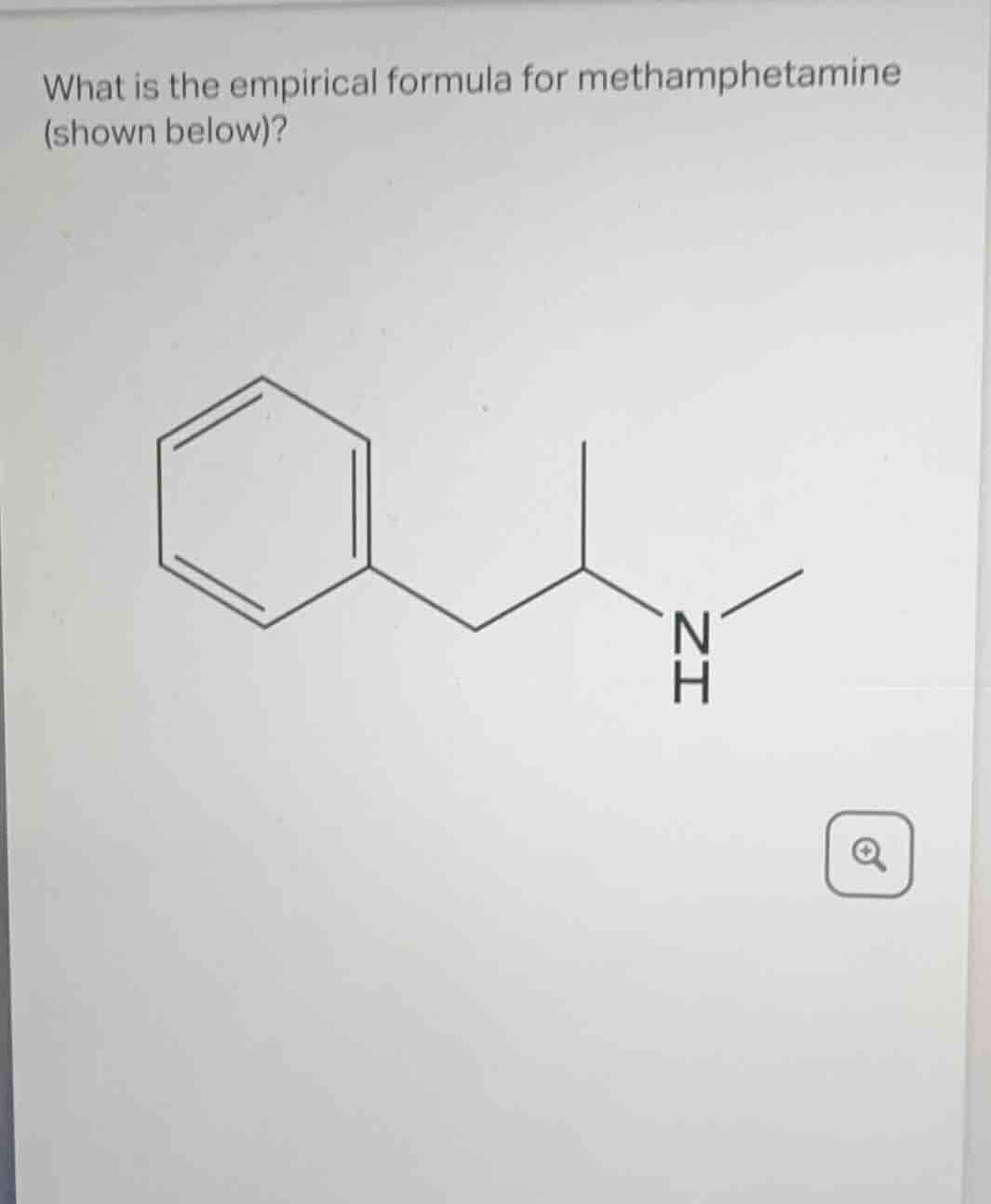
what is the empirical formula for methamphetamine (shown below)?
Step1: Count atoms in structure
The structure has a benzene ring (6 C), a -CH₂- (1 C), a -CH- (1 C), a -NH- (1 N, 1 H), and two -CH₃-? Wait, no, looking at the structure: benzene ring (C₆H₅), then -CH₂-CH(CH₃)-NH-CH₃? Wait, no, the structure is: benzene attached to -CH₂-CH(NHCH₃)-? Wait, no, the drawing: benzene ring, then a -CH₂- (so C₇), then a -CH- (C₈), then attached to N with a -CH₃ (so N, H, and CH₃: N, H, C). Wait, let's count properly:
Benzene ring: 6 C, 5 H (since it's a phenyl group, C₆H₅). Then the chain: -CH₂- (C₇, H₂), -CH- (C₈, H), then N with H and a -CH₃ (N, H, C₉, H₃). Wait, no, the structure is:
Phenyl (C₆H₅) - CH₂ - CH(NHCH₃). Wait, the N is attached to the CH and to a CH₃. So let's list all atoms:
Carbon: benzene (6) + CH₂ (1) + CH (1) + CH₃ (1) = 6+1+1+1=9? Wait, no, the CH is attached to N and to CH₂, and N is attached to CH₃. So:
C: 6 (benzene) + 1 (CH₂) + 1 (CH) + 1 (CH₃) = 9
H: benzene (5) + CH₂ (2) + CH (1) + NH (1) + CH₃ (3) = 5+2+1+1+3=12? Wait, no: benzene is C₆H₅ (5 H), CH₂ has 2 H, CH has 1 H, NH has 1 H, CH₃ has 3 H. So total H: 5+2+1+1+3=12? Wait, no, maybe I messed up. Wait, the structure is:
Phenyl (C₆H₅) - CH₂ - CH(NHCH₃). So the CH is bonded to CH₂, to N, and to... wait, the CH has one bond to CH₂, one to N, and one to...? Wait, no, the drawing: the CH is a carbon with three bonds: to CH₂, to N, and to...? Wait, no, the structure is:
Benzene ring (C₆H₅) connected to -CH₂- (C₇H₂), then -CH- (C₈H) connected to N (NH) and to a -CH₃ (C₉H₃). So N is bonded to CH (C₈) and to CH₃ (C₉). So:
C: 6 (benzene) + 1 (CH₂) + 1 (CH) + 1 (CH₃) = 9
H: benzene (5) + CH₂ (2) + CH (1) + NH (1) + CH₃ (3) = 5+2+1+1+3=12? Wait, no, NH is N-H, so H from NH is 1, CH₃ is 3 H, CH has 1 H, CH₂ has 2 H, benzene has 5 H. So total H: 5+2+1+1+3=12? Wait, no, maybe the CH is bonded to CH₂, N, and a methyl? Wait, no, the structure is:
The correct molecular formula for methamphetamine is C₁₀H₁₅N? Wait, no, wait, methamphetamine's molecular formula is C₁₀H₁₅N? Wait, no, let's check: methamphetamine is (S)-N-methyl-1-phenylpropan-2-amine, which has formula C₁₀H₁₅N. Wait, so empirical formula is the simplest ratio. C₁₀H₁₅N: divide by GCD(10,15,1)=5? No, GCD(10,15,1) is 1? Wait, 10,15,1: GCD is 1. Wait, no, wait, 10 and 15 have GCD 5. Wait, 10/5=2, 15/5=3, 1/1=1. So empirical formula would be C₂H₃N? No, wait, no, wait, methamphetamine's molecular formula is C₁₀H₁₅N. So to find empirical formula, we divide each subscript by the greatest common divisor (GCD) of 10,15,1. The GCD of 10,15,1 is 1. Wait, no, 10 and 15 have GCD 5, but N is 1, so GCD is 1. Wait, that can't be. Wait, maybe I counted wrong. Wait, let's check the structure again.
Wait, the structure in the image: benzene ring (C₆H₅), then -CH₂-CH(NHCH₃). Wait, the CH is attached to N and to a CH₃, and N is attached to H. So the atoms:
C: 6 (benzene) + 1 (CH₂) + 1 (CH) + 1 (CH₃) + 1 (NHCH₃? No, N is attached to CH and to CH₃. Wait, no, the correct structure of methamphetamine is: C₆H₅ - CH₂ - CH(NHCH₃). Wait, no, it's C₆H₅ - CH₂ - CH(NHCH₃) is wrong. The correct structure is C₆H₅ - CH₂ - CH(CH₃) - NHCH₃. Wait, that's C₁₀H₁₅N. Let's count:
C: 6 (benzene) + 1 (CH₂) + 1 (CH) + 1 (CH₃) + 1 (CH₃) = 6+1+1+1+1=10? Wait, no, CH₂, CH, and two CH₃? Wait, no, methamphetamine is N-methyl-1-phenylpropan-2-amine, which is:
Phenyl (C₆H₅) - CH₂ - CH(CH₃) - NH - CH₃. So that's:
C: 6 (phenyl) + 1 (CH₂) + 1 (CH) + 1 (CH₃) + 1 (CH₃) = 6+1+1+1+1=10
H: 5 (phenyl) + 2 (CH₂) + 1 (CH) + 3 (CH₃) + 1 (NH) + 3 (CH₃) = 5+2+1+3+1+3=15
N: 1
So molecular formula is C₁₀H₁₅N. Now, to find empiri…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boxed{C_{10}H_{15}N}$