QUESTION IMAGE
Question
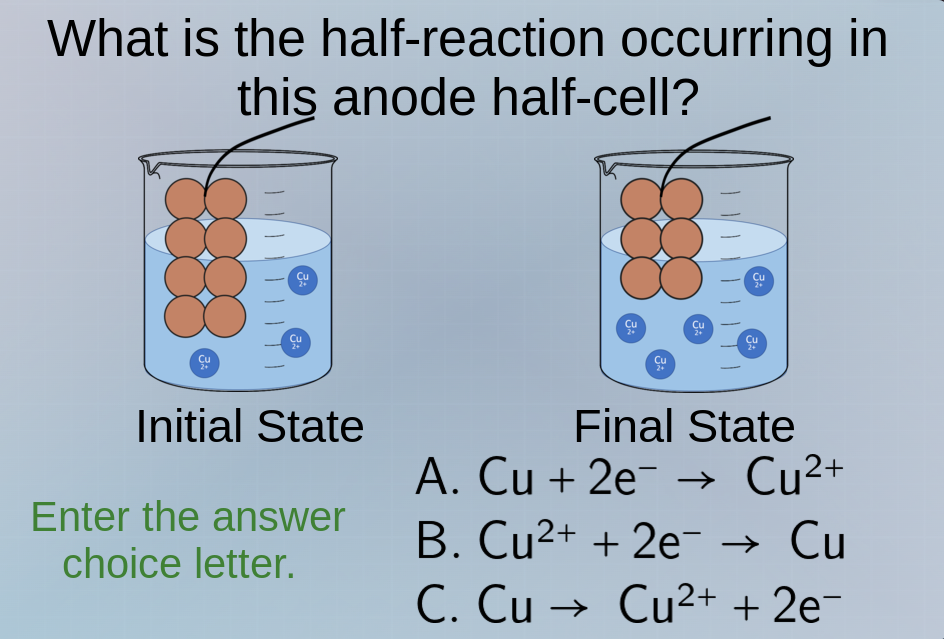
what is the half - reaction occurring in this anode half - cell? initial state final state enter the answer choice letter. a. $\ce{cu + 2e^- -> cu^{2+}}$ b. $\ce{cu^{2+} + 2e^- -> cu}$ c. $\ce{cu -> cu^{2+} + 2e^-}$
At the anode, oxidation occurs (loss of electrons). In the initial to final state, solid Cu is converting to \(Cu^{2+}\) ions, meaning Cu is losing electrons. Option A is reduction (gaining electrons, wrong direction), Option B is reduction (gain of electrons, cathode reaction), Option C shows Cu losing 2 electrons to form \(Cu^{2+}\), which is oxidation at the anode.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \( \text{Cu}
ightarrow \text{Cu}^{2+} + 2\text{e}^{-} \)