QUESTION IMAGE
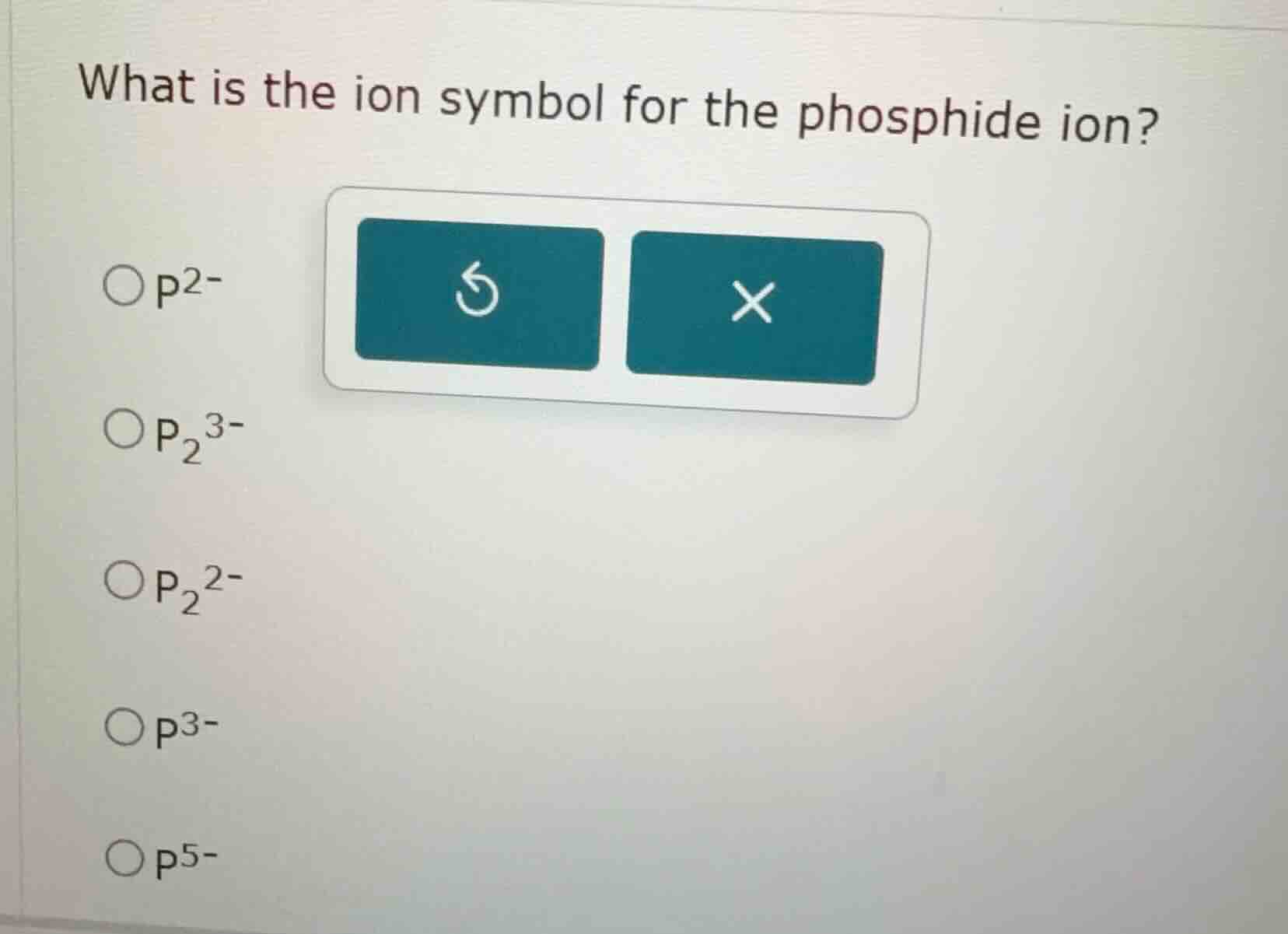
Question
what is the ion symbol for the phosphide ion?
options:
- $\ce{p^{2-}}$
- $\ce{p2^{3-}}$
- $\ce{p2^{2-}}$
- $\ce{p^{3-}}$
- $\ce{p^{5-}}$
Phosphorus (P) is in group 15 of the periodic table. Atoms in group 15 typically gain 3 electrons to achieve a stable electron configuration, forming an anion with a 3 - charge. The phosphide ion is a single phosphorus atom with a 3 - charge, so its symbol is $P^{3 - }$ (note: the correct symbol uses a capital "P" for phosphorus, and the charge is 3 - ). Among the options, $P^{3 - }$ (or written as p³⁻ with lowercase, but element symbols should have the first letter capitalized; however, looking at the options, the last option with p³⁻ is the intended correct one as it represents the phosphide ion with a 3 - charge from a single P atom).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $p^{3 - }$ (assuming the last option is labeled D, with the option text $p^{3 - }$)