QUESTION IMAGE
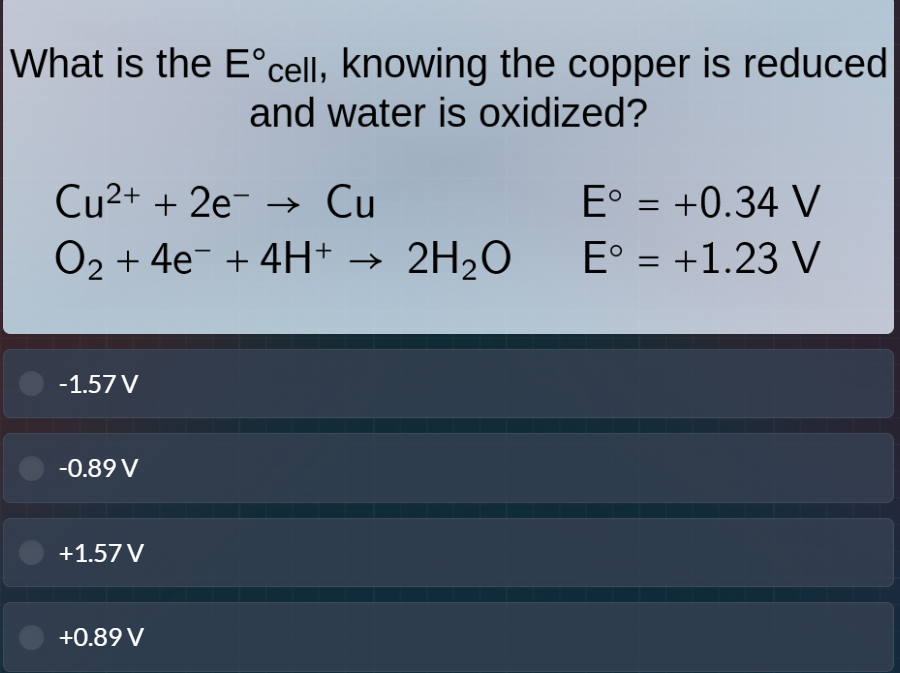
Question
what is the $\text{e}^\circ_{\text{cell}}$, knowing the copper is reduced and water is oxidized?
$\text{cu}^{2+} + 2\text{e}^- \
ightarrow \text{cu}$ \quad \quad \quad \quad $\text{e}^\circ = +0.34$ v
$\text{o}_2 + 4\text{e}^- + 4\text{h}^+ \
ightarrow 2\text{h}_2\text{o}$ \quad $\text{e}^\circ = +1.23$ v
-1.57v
-0.89v
+1.57v
+0.89v
Step1: Identify reduction and oxidation half - reactions
Reduction: \(Cu^{2 +}+2e^-
ightarrow Cu\), \(E_{red}^o = + 0.34\ V\)
Oxidation: The reverse of \(O_2 + 4e^-+4H^+
ightarrow2H_2O\) is \(2H_2O
ightarrow O_2 + 4e^-+4H^+\), so \(E_{ox}^o=- 1.23\ V\) (since oxidation is reverse of reduction, we take the negative of the reduction potential)
Step2: Balance electrons
To balance electrons, multiply the reduction half - reaction by 2: \(2Cu^{2 +}+4e^-
ightarrow2Cu\), \(E_{red}^o = + 0.34\ V\) (the potential does not change with multiplication of stoichiometric coefficients)
The oxidation half - reaction is \(2H_2O
ightarrow O_2 + 4e^-+4H^+\), \(E_{ox}^o=-1.23\ V\)
Step3: Calculate \(E^o_{cell}\)
The formula for \(E^o_{cell}\) is \(E^o_{cell}=E^o_{red}(cathode)+E^o_{ox}(anode)\)
Substitute the values: \(E^o_{cell}=0.34\ V+( - 1.23\ V)=0.34 - 1.23=- 0.89\ V\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 0.89 V