QUESTION IMAGE
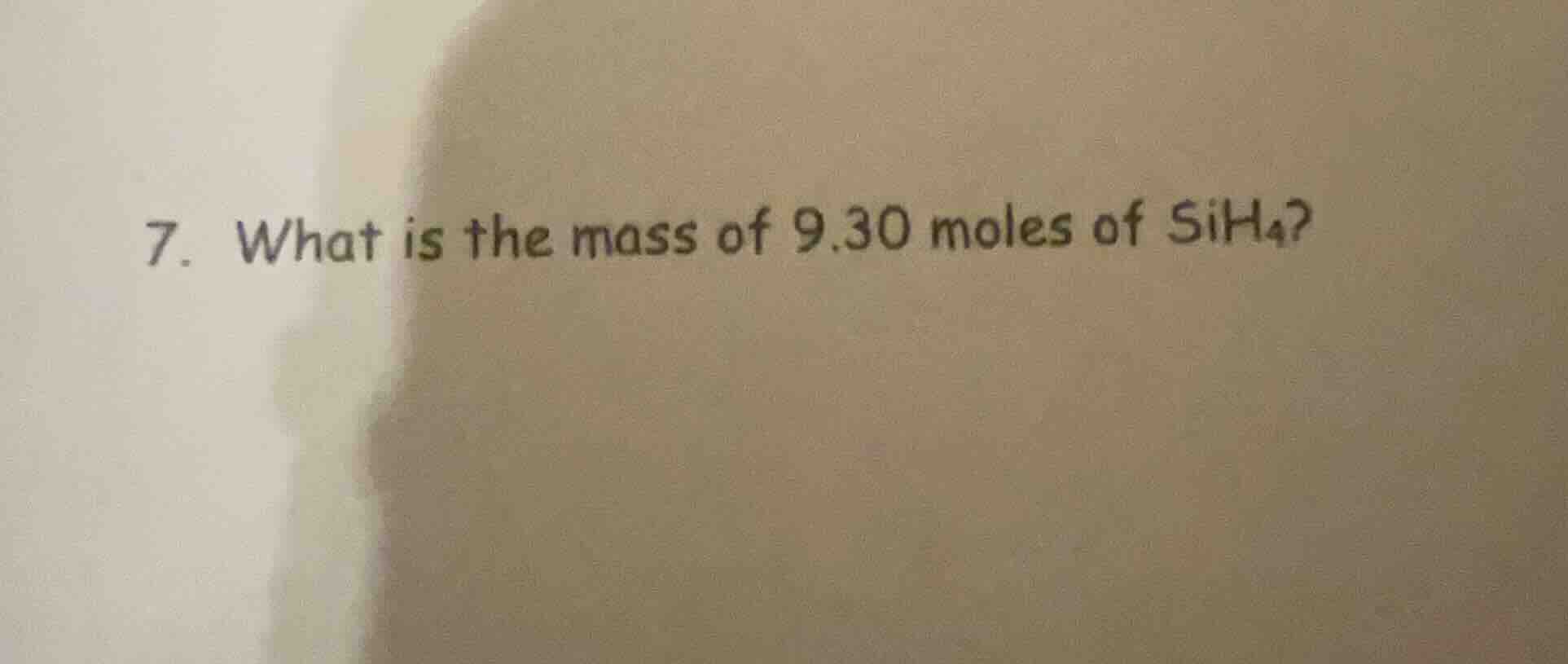
Question
- what is the mass of 9.30 moles of sih₄?
Step1: Calculate molar mass of $\text{SiH}_4$
Molar mass of $\text{Si} = 28.09\ \text{g/mol}$, molar mass of $\text{H} = 1.008\ \text{g/mol}$
$$\text{Molar mass of }\text{SiH}_4 = 28.09 + 4\times1.008 = 32.122\ \text{g/mol}$$
Step2: Compute mass from moles
Use $\text{Mass} = \text{Moles} \times \text{Molar Mass}$
$$\text{Mass} = 9.30\ \text{mol} \times 32.122\ \text{g/mol}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$298.7346\ \text{g}$ (or rounded to $299\ \text{g}$ for 3 significant figures)