QUESTION IMAGE
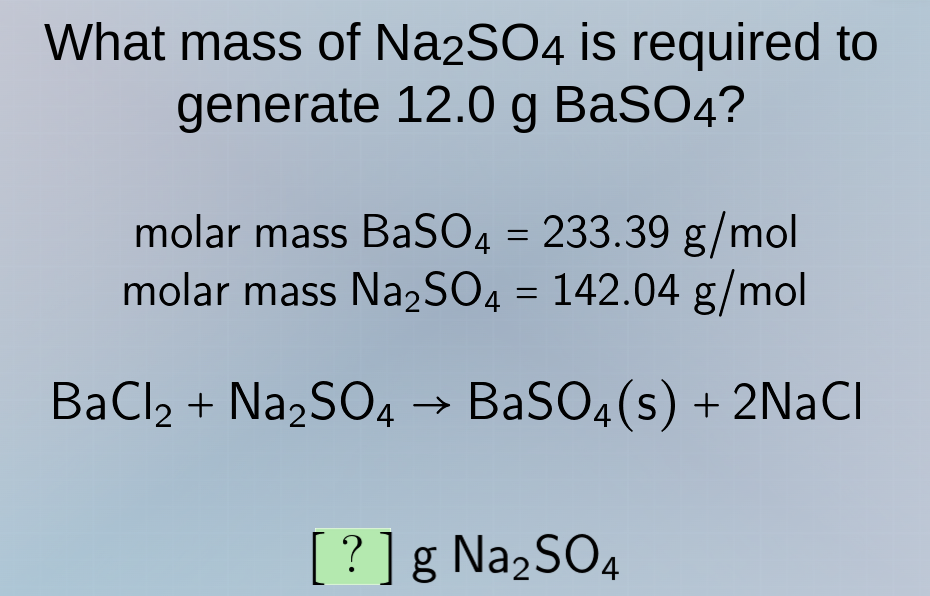
Question
what mass of na₂so₄ is required to generate 12.0 g baso₄? molar mass baso₄ = 233.39 g/mol molar mass na₂so₄ = 142.04 g/mol bacl₂ + na₂so₄ → baso₄(s) + 2nacl ? g na₂so₄
Step1: Calculate moles of BaSO₄
Moles of a substance are calculated by dividing its mass by its molar mass. The formula for moles ($n$) is $n=\frac{m}{M}$, where $m$ is the mass and $M$ is the molar mass.
For BaSO₄, $m = 12.0\space g$ and $M = 233.39\space g/mol$.
So, moles of BaSO₄, $n_{BaSO_4}=\frac{12.0\space g}{233.39\space g/mol}\approx0.05142\space mol$.
Step2: Determine moles of Na₂SO₄ from stoichiometry
From the balanced chemical equation $BaCl_2 + Na_2SO_4
ightarrow BaSO_4(s) + 2NaCl$, the mole ratio of $Na_2SO_4$ to $BaSO_4$ is 1:1.
This means moles of $Na_2SO_4$ ($n_{Na_2SO_4}$) is equal to moles of $BaSO_4$. So, $n_{Na_2SO_4}=n_{BaSO_4}\approx0.05142\space mol$.
Step3: Calculate mass of Na₂SO₄
Using the formula $m = n\times M$, where $n$ is the moles of $Na_2SO_4$ and $M$ is its molar mass.
For $Na_2SO_4$, $n = 0.05142\space mol$ and $M = 142.04\space g/mol$.
So, mass of $Na_2SO_4$, $m_{Na_2SO_4}=0.05142\space mol\times142.04\space g/mol\approx7.30\space g$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\boxed{7.30}