QUESTION IMAGE
Question
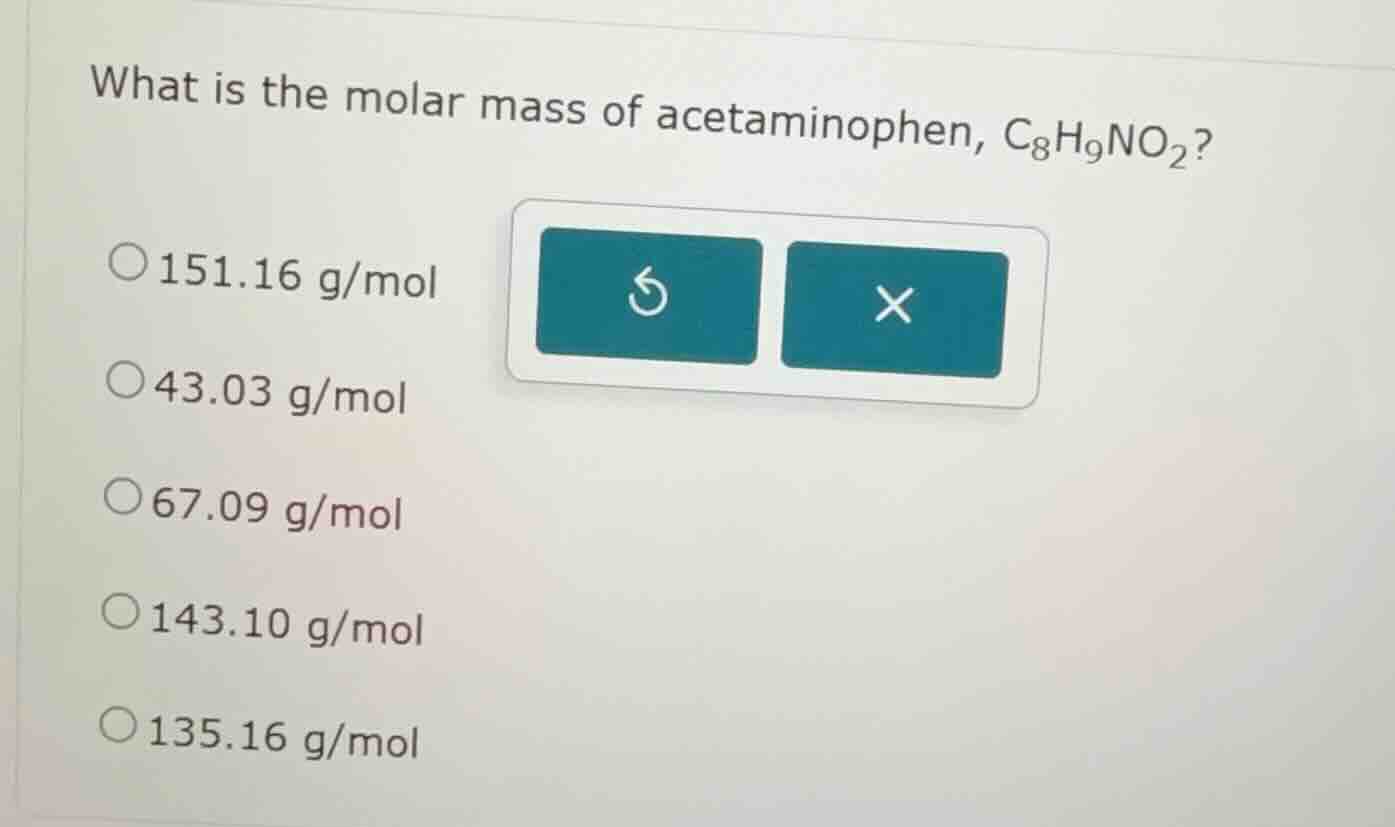
what is the molar mass of acetaminophen, c₈h₉no₂?
○151.16 g/mol
○43.03 g/mol
○67.09 g/mol
○143.10 g/mol
○135.16 g/mol
Step1: Recall atomic masses
Atomic masses: C = 12.01 g/mol, H = 1.008 g/mol, N = 14.01 g/mol, O = 16.00 g/mol.
Step2: Calculate for each element
For C: \( 8 \times 12.01 = 96.08 \)
For H: \( 9 \times 1.008 = 9.072 \)
For N: \( 1 \times 14.01 = 14.01 \)
For O: \( 2 \times 16.00 = 32.00 \)
Step3: Sum all contributions
Total molar mass = \( 96.08 + 9.072 + 14.01 + 32.00 \)
\( = 96.08 + 9.072 = 105.152 \); \( 105.152 + 14.01 = 119.162 \); \( 119.162 + 32.00 = 151.162 \approx 151.16 \) g/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
151.16 g/mol