QUESTION IMAGE
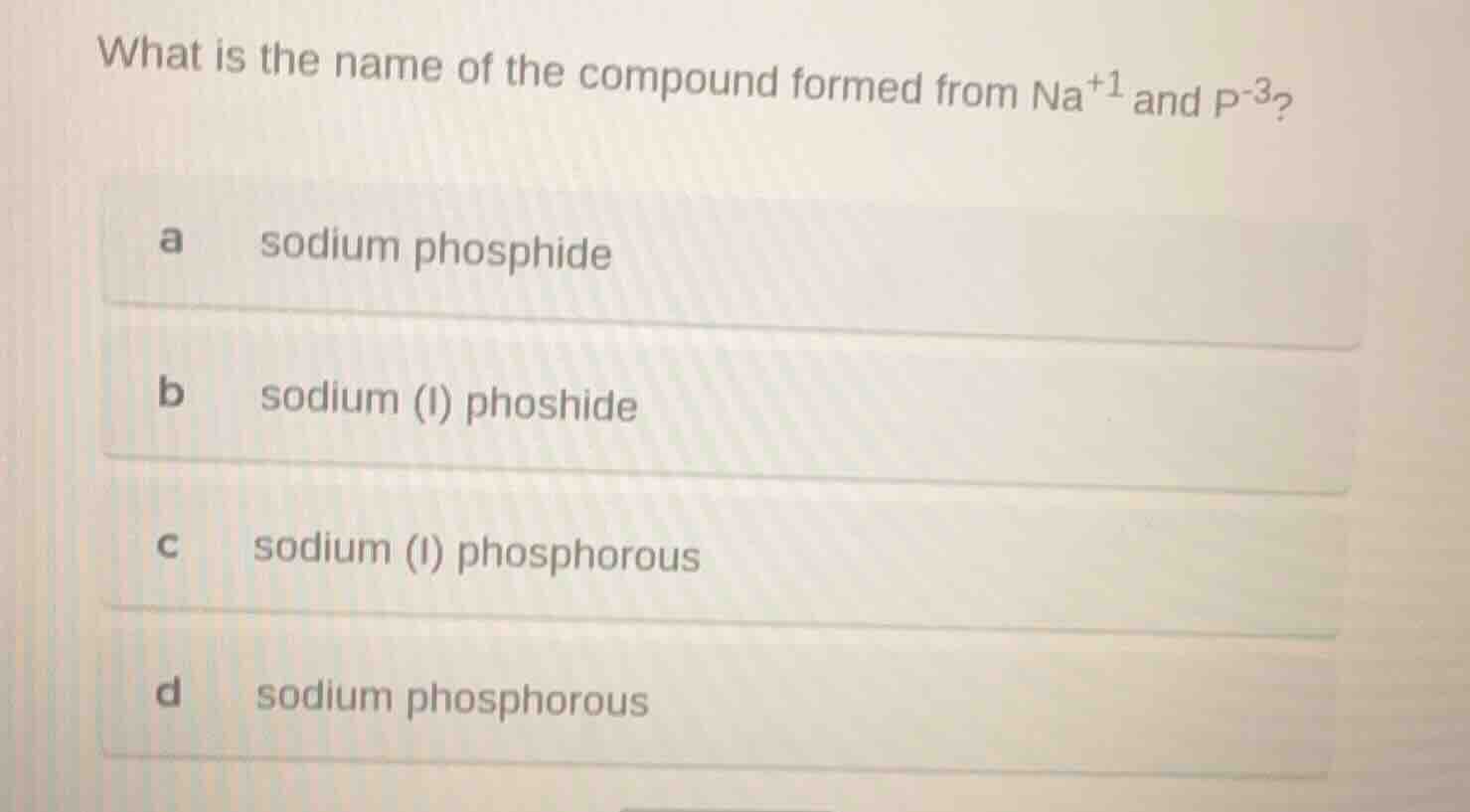
Question
what is the name of the compound formed from na⁺¹ and p⁻³?
a sodium phosphide
b sodium (i) phoshide
c sodium (i) phosphorous
d sodium phosphorous
To determine the compound name, first find the formula from ions \( \text{Na}^{+1} \) and \( \text{P}^{-3} \). Using the criss - cross method, the formula is \( \text{Na}_3\text{P} \). For ionic compounds with a metal (sodium, which has a fixed charge of +1) and a non - metal (phosphide ion \( \text{P}^{-3} \)), we name the cation (sodium) first, then the anion (phosphide). Sodium has a fixed oxidation state, so we don't use a Roman numeral. Option b has a misspelling (“phoshide”) and incorrect use of Roman numeral. Options c and d use “phosphorous” which is incorrect (the anion is phosphide, and “phosphorous” refers to the element in a different context or to a different ion like phosphite/phosphate - related, not the \( \text{P}^{-3} \) anion). So the correct name is sodium phosphide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. sodium phosphide