QUESTION IMAGE
Question
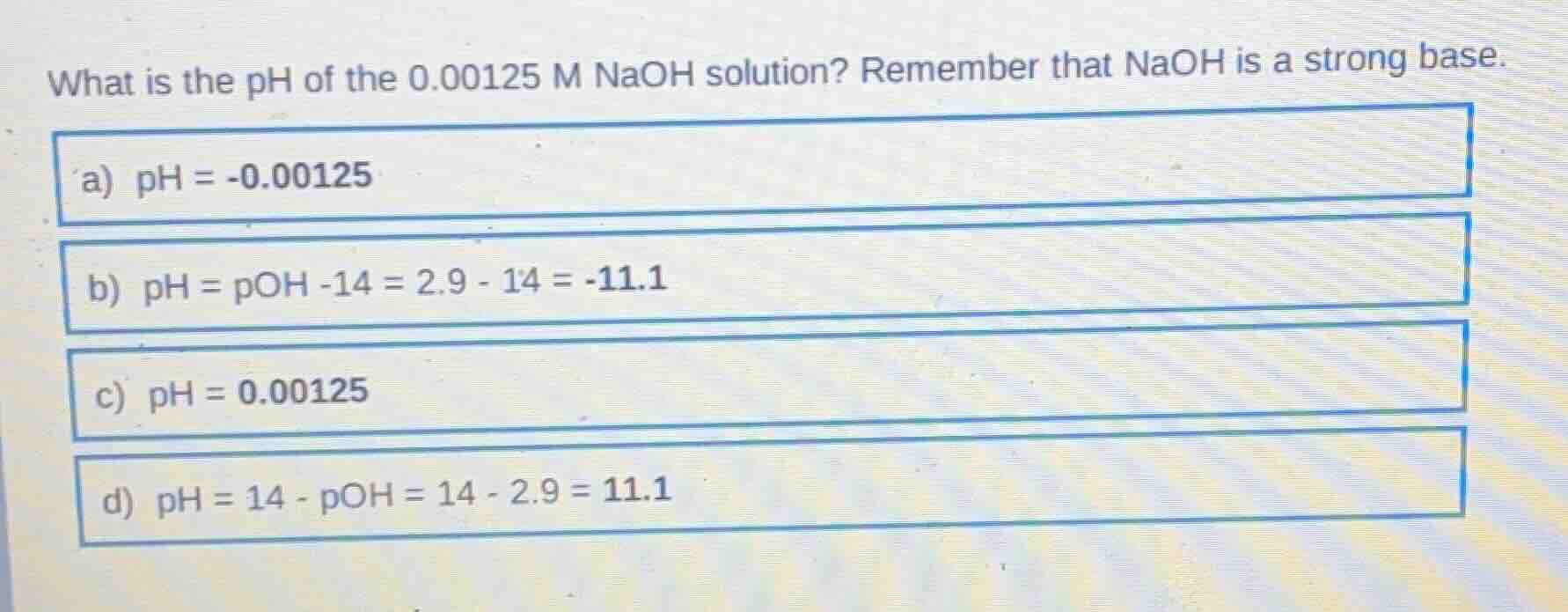
what is the ph of the 0.00125 m naoh solution? remember that naoh is a strong base. a) ph = -0.00125 b) ph = poh -14 = 2.9 - 14 = -11.1 c) ph = 0.00125 d) ph = 14 - poh = 14 - 2.9 = 11.1
Step1: Find $[\text{OH}^-]$ concentration
Since NaOH is a strong base, it dissociates completely: $\text{NaOH}
ightarrow \text{Na}^+ + \text{OH}^-$, so $[\text{OH}^-] = 0.00125\ \text{M}$.
Step2: Calculate pOH from $[\text{OH}^-]$
$\text{pOH} = -\log_{10}[\text{OH}^-] = -\log_{10}(0.00125) \approx 2.9$
Step3: Calculate pH from pOH
Use the relation $\text{pH} + \text{pOH} = 14$, so rearrange to $\text{pH} = 14 - \text{pOH}$.
$\text{pH} = 14 - 2.9 = 11.1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d) $\text{pH} = 14 - \text{pOH} = 14 - 2.9 = 11.1$