QUESTION IMAGE
Question
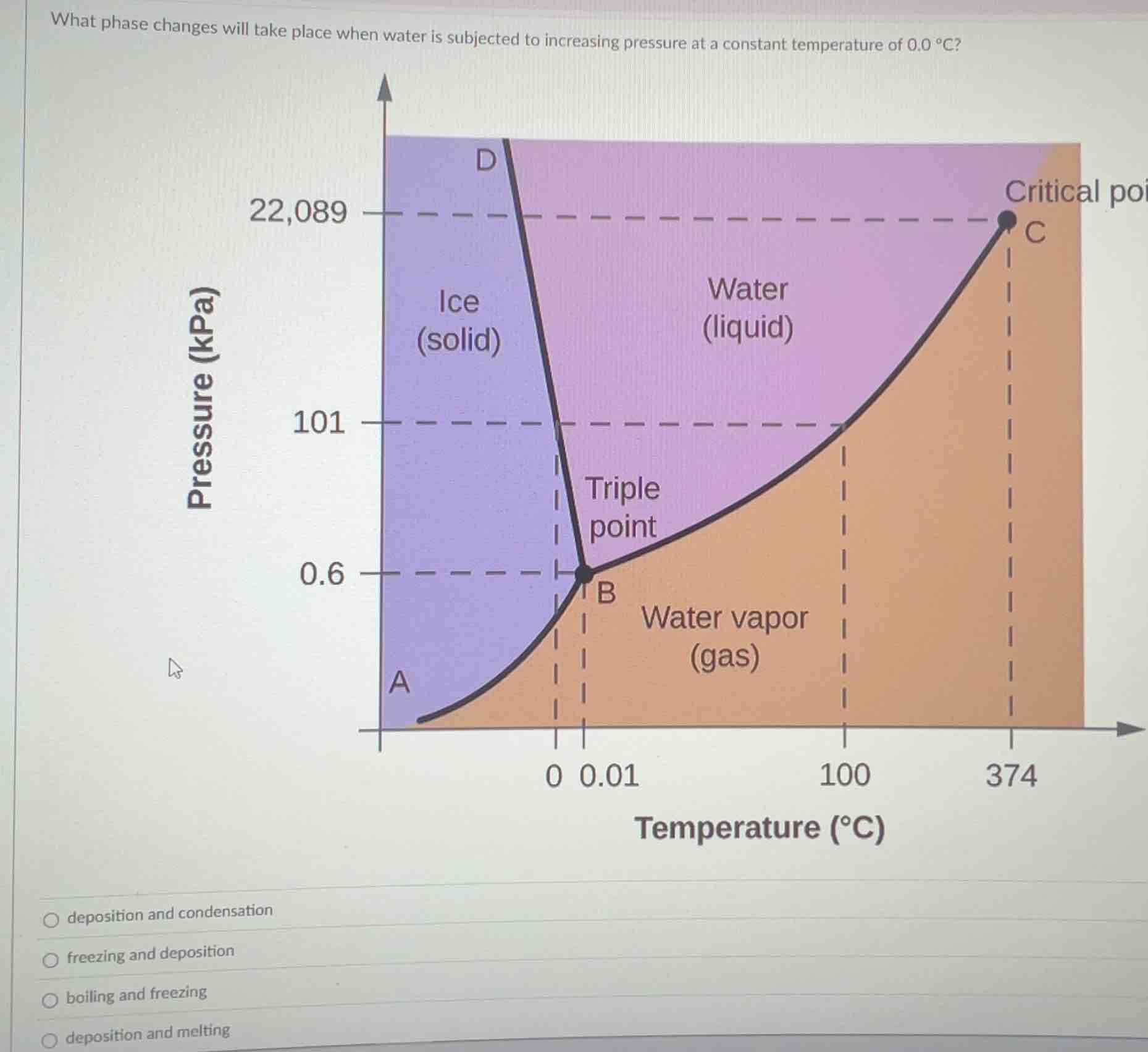
what phase changes will take place when water is subjected to increasing pressure at a constant temperature of 0.0 °c?
phase diagram of water with pressure (kpa) on y - axis and temperature (°c) on x - axis, regions for ice (solid), water (liquid), water vapor (gas), triple point, critical point, and labeled curves. below the diagram: multiple - choice options:
- deposition and condensation
- freezing and deposition
- boiling and freezing
- deposition and melting
Brief Explanations
- First, analyze the phase diagram of water. At 0.0 °C (constant temperature), when pressure increases:
- For water vapor (gas) at low pressure, increasing pressure at constant temperature (0.0 °C) will cause it to change to solid (ice) first? No, wait, let's check the regions. The triple point is at 0.01 °C and 0.6 kPa. At 0.0 °C, which is slightly below the triple - point temperature? Wait, no, 0.0 °C is above the triple - point temperature (0.01 °C)? Wait, the triple - point temperature of water is 0.01 °C. So at 0.0 °C (close to triple - point), when pressure increases:
- If we start from water vapor (gas) region, increasing pressure at constant temperature (0.0 °C) will first cause deposition (gas to solid) if pressure is low, but then when pressure crosses the ice - liquid boundary? Wait, no, the key is to look at the phase boundaries. The line between ice (solid) and water vapor (gas) is curve A - B. The line between ice (solid) and liquid water is the left - most curve (D - B? Wait, the diagram: the purple region is ice (solid), pink is liquid water, orange is water vapor (gas).
- At constant temperature of 0.0 °C, as pressure increases:
- If we are in the water vapor (gas) region (low pressure), increasing pressure will first cause deposition (gas→solid, because the boundary between gas and solid is crossed). Then, as pressure further increases, crossing the solid - liquid boundary (the left - hand curve), the solid (ice) will melt into liquid water (melting: solid→liquid). So the phase changes are deposition (gas→solid) and melting (solid→liquid).
- Now let's analyze the options:
- Option 1: deposition and condensation. Condensation is gas→liquid, but at 0.0 °C, to get to liquid from gas, we need to cross the gas - liquid boundary, but the gas - liquid boundary at 0.0 °C is at higher pressure? Wait, no, the gas - liquid boundary (the curve from triple point to critical point) at 0.0 °C is at pressure higher than 0.6 kPa? Wait, the triple point is at 0.01 °C and 0.6 kPa. At 0.0 °C (slightly lower than triple - point temperature), the gas - solid boundary and solid - liquid boundary: when pressure increases from very low (in gas region) at 0.0 °C, first gas→solid (deposition), then solid→liquid (melting).
- Option 2: freezing and deposition. Freezing is liquid→solid, but we start from gas or maybe liquid? No, at 0.0 °C, if we start from liquid, increasing pressure would not cause freezing (water expands when it freezes, so increasing pressure at 0.0 °C on liquid water would not cause freezing, in fact, it would stay liquid or melt ice). So freezing is wrong.
- Option 3: boiling and freezing. Boiling is liquid→gas, which is opposite of what happens when pressure increases. Freezing is also wrong.
- Option 4: deposition and melting. Deposition (gas→solid) and melting (solid→liquid) match our analysis.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
deposition and melting