QUESTION IMAGE
Question
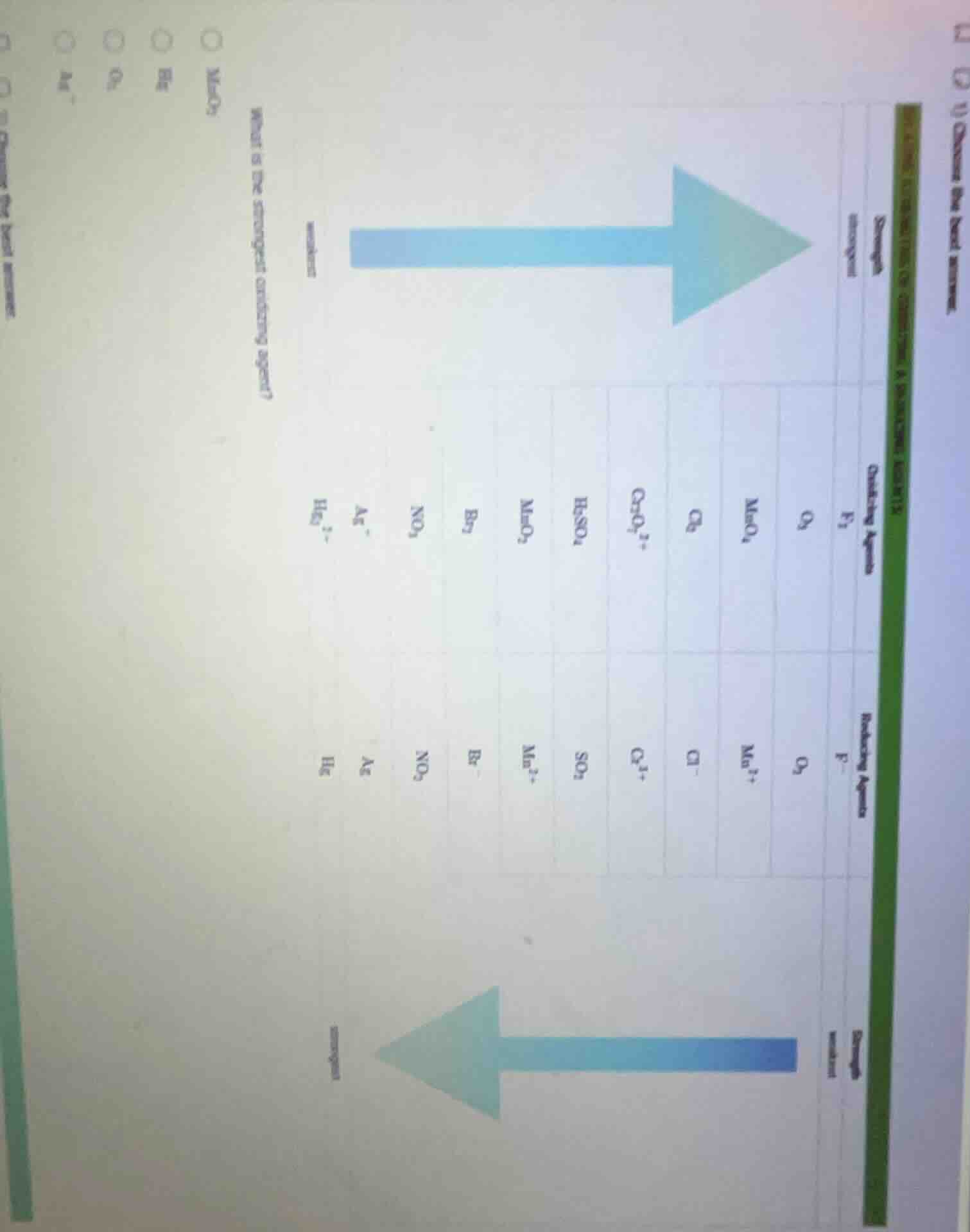
what is the strongest oxidizing agent?
oxidizing agents: f₂, o₂, mno₄⁻, cl₂, cr₂o₇²⁻, h₂so₄, mno₂, br₂, no₃⁻, ag⁺, hg₂²⁺
reducing agents: f⁻, o₂, mn²⁺, cl⁻, cr³⁺, so₃²⁻, mn²⁺, br⁻, no₂⁻, ag, hg
The provided chart shows that oxidizing agent strength increases in the direction of the top arrow. The leftmost oxidizing agent $\text{F}_2$ is the strongest, but it is not an option. Looking at the given options, $\text{MnO}_2$ is positioned further to the right (stronger) than $\text{Hg}$, $\text{O}_2$, and $\text{Ag}^+$ in the oxidizing agents list, making it the strongest among the choices.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{MnO}_2$