QUESTION IMAGE
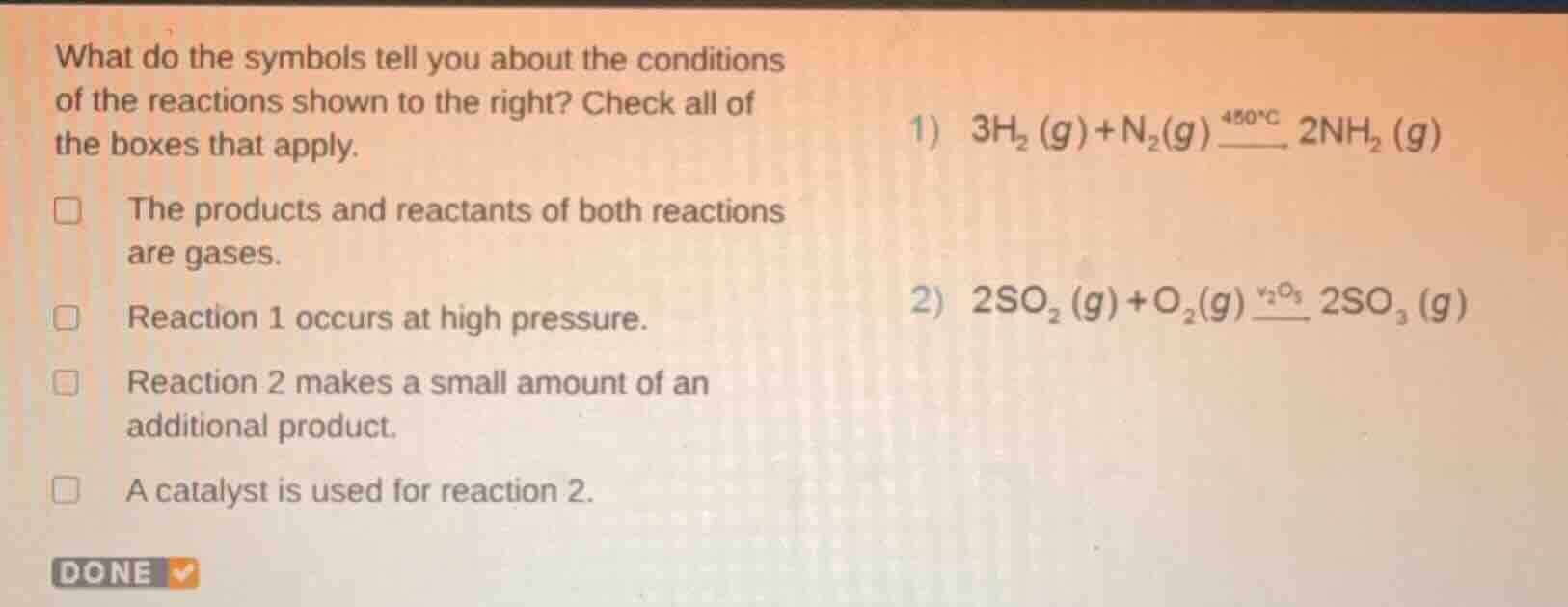
Question
what do the symbols tell you about the conditions of the reactions shown to the right? check all of the boxes that apply. the products and reactants of both reactions are gases. reaction 1 occurs at high pressure. reaction 2 makes a small amount of an additional product. a catalyst is used for reaction 2. 1) \\( 3h_2 (g)+n_2(g) \stackrel{450^circ c}{\\_\\_\\_\\_} 2nh_2 (g) \\) 2) \\( 2so_2 (g)+o_2(g) \stackrel{v_2o_3}{\\_\\_\\_\\_} 2so_3 (g) \\)
Brief Explanations
- For the state symbols: In both reactions, reactants and products have the (g) symbol, meaning they are gases. So "The products and reactants of both reactions are gases" is correct.
- Reaction 1: The reaction for ammonia synthesis (Haber process) typically occurs at high pressure, and the reaction is written with conditions suggesting industrial process conditions, so "Reaction 1 occurs at high pressure" is correct (implied by industrial context of \(3H_2(g)+N_2(g)\) to \(2NH_3(g)\) at \(450^\circ C\), which uses high pressure).
- Reaction 2: The symbol \(V_2O_5\) above the arrow is a catalyst. There's no indication of additional product, so "A catalyst is used for reaction 2" is correct, and "Reaction 2 makes a small amount of an additional product" is incorrect.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The products and reactants of both reactions are gases.
- Reaction 1 occurs at high pressure.
- A catalyst is used for reaction 2.