QUESTION IMAGE
Question
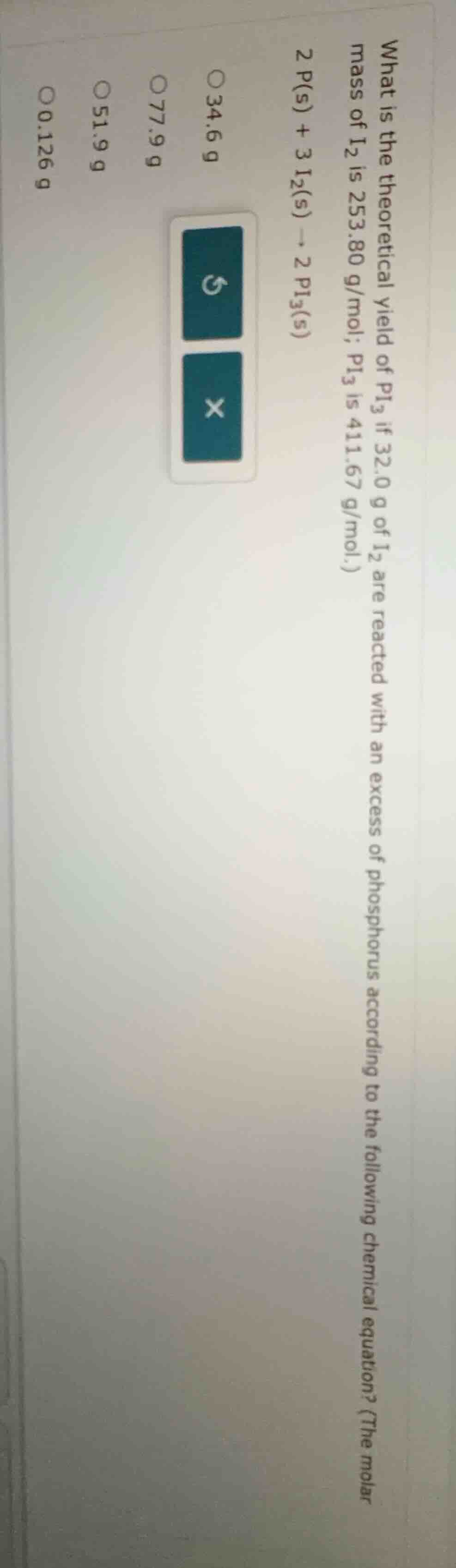
what is the theoretical yield of pi₃ if 32.0 g of i₂ are reacted with an excess of phosphorus according to the following chemical equation? (the molar mass of i₂ is 253.80 g/mol; pi₃ is 411.67 g/mol.)
2 p(s) + 3 i₂(s) → 2 pi₃(s)
34.6 g
77.9 g
51.9 g
0.126 g
Step1: Calculate moles of \( I_2 \)
Moles of \( I_2 = \frac{\text{mass of } I_2}{\text{molar mass of } I_2} = \frac{32.0\ \text{g}}{253.80\ \text{g/mol}} \approx 0.126\ \text{mol} \)
Step2: Determine mole ratio of \( I_2 \) to \( PI_3 \)
From the equation \( 2P + 3I_2
ightarrow 2PI_3 \), the ratio of \( I_2 \) to \( PI_3 \) is \( 3:2 \).
Moles of \( PI_3 = \frac{2}{3} \times \) moles of \( I_2 = \frac{2}{3} \times 0.126\ \text{mol} = 0.084\ \text{mol} \)
Step3: Calculate mass of \( PI_3 \)
Mass of \( PI_3 = \text{moles of } PI_3 \times \text{molar mass of } PI_3 = 0.084\ \text{mol} \times 411.67\ \text{g/mol} \approx 34.6\ \text{g} \)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
34.6 g