QUESTION IMAGE
Question
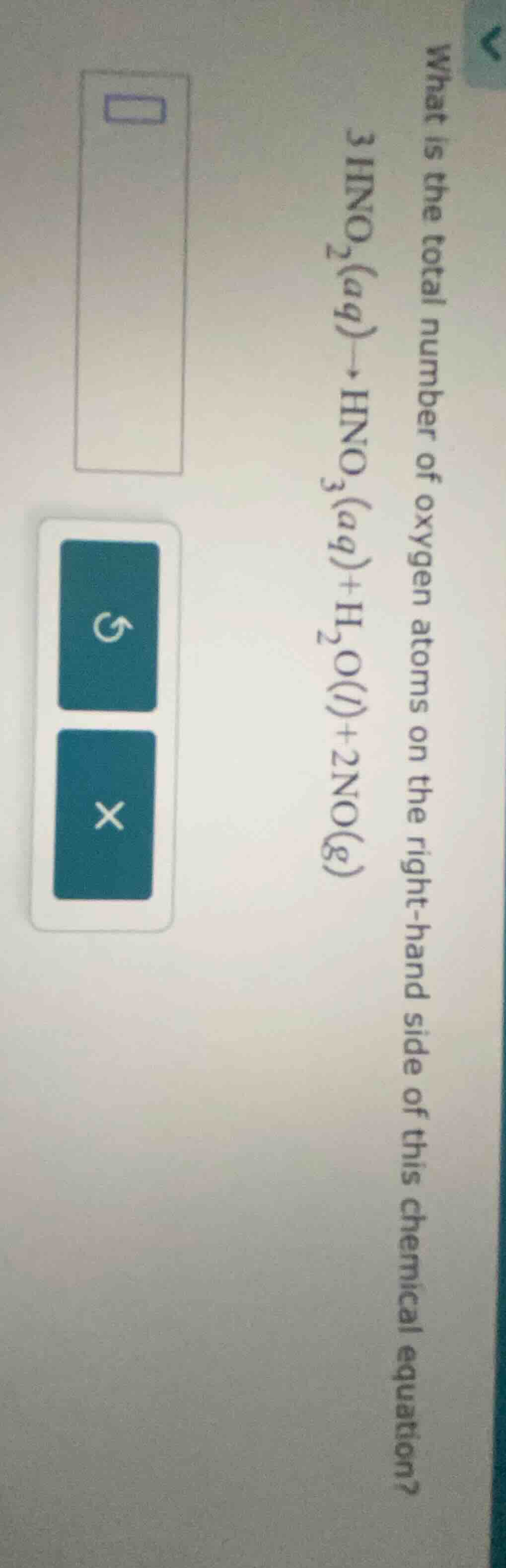
what is the total number of oxygen atoms on the right - hand side of this chemical equation?
$3\ce{hno_{2}(aq)\
ightarrow hno_{3}(aq) + h_{2}o(l) + 2no(g)}$
Step1: Analyze \( \text{HNO}_3 \)
In \( \text{HNO}_3 \), the number of O atoms is 3.
Step2: Analyze \( \text{H}_2\text{O} \)
In \( \text{H}_2\text{O} \), the number of O atoms is 1.
Step3: Analyze \( 2\text{NO} \)
In \( \text{NO} \), the number of O atoms is 1, and for \( 2\text{NO} \), it is \( 2\times1 = 2 \).
Step4: Sum the O atoms
Total O atoms \( = 3 + 1 + 2 = 6 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6