QUESTION IMAGE
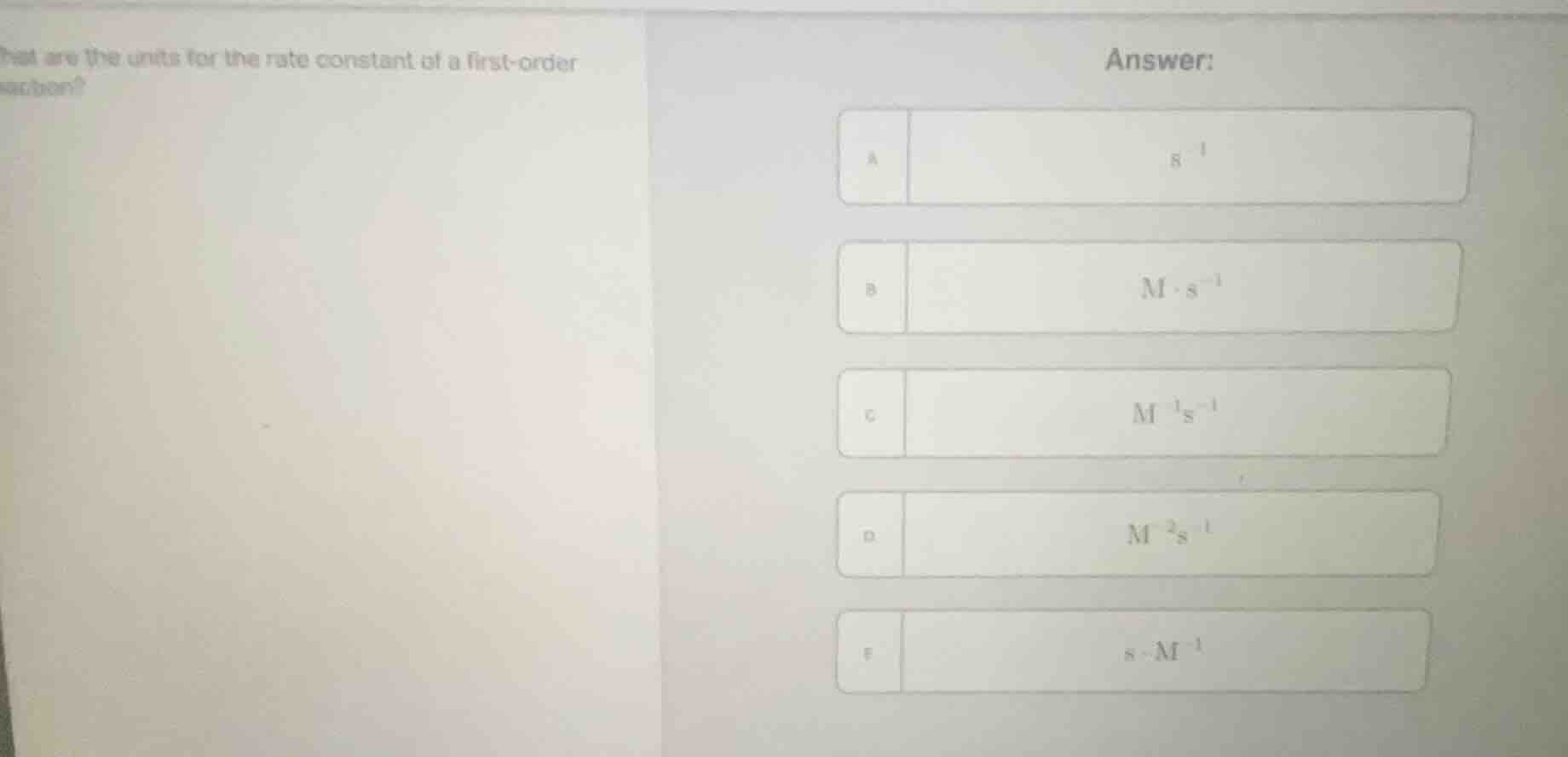
Question
what are the units for the rate constant of a first-order reaction?
answer:
a $mathrm{s^{-1}}$
b $mathrm{mcdot s^{-1}}$
c $mathrm{m^{-1}s^{-1}}$
d $mathrm{m^{-2}s^{-1}}$
e $mathrm{scdot m^{-1}}$
Step1: Recall first-order rate law
The rate law for a first-order reaction is:
$$\text{Rate} = k[A]$$
where $\text{Rate}$ has units of concentration per time ($\text{M} \cdot \text{s}^{-1}$), $[A]$ is concentration ($\text{M}$), and $k$ is the rate constant.
Step2: Solve for units of $k$
Rearrange to isolate $k$ and substitute units:
$$k = \frac{\text{Rate}}{[A]} = \frac{\text{M} \cdot \text{s}^{-1}}{\text{M}}$$
Step3: Simplify the units
Cancel the $\text{M}$ terms:
$$k = \text{s}^{-1}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\text{s}^{-1}$