QUESTION IMAGE
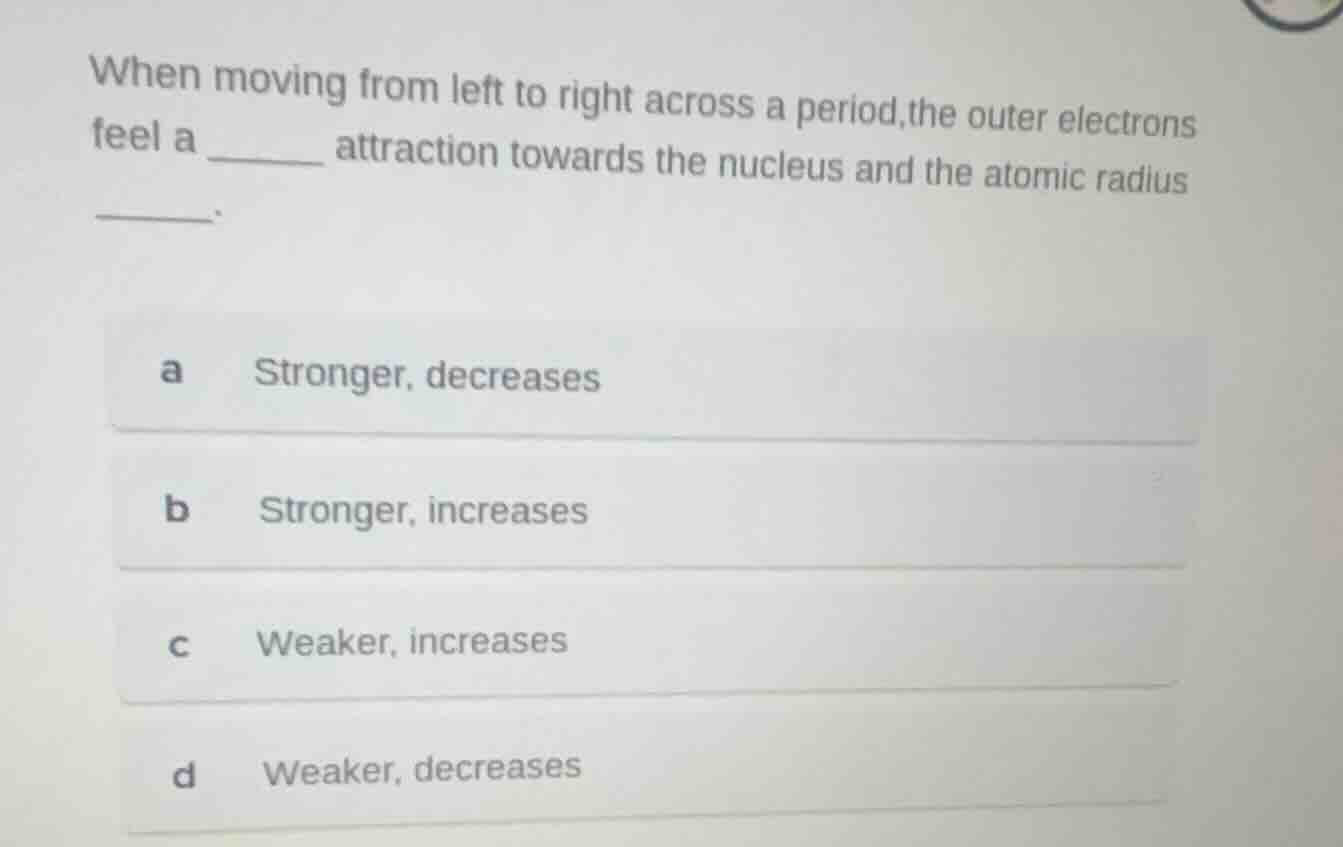
Question
when moving from left to right across a period,the outer electrons feel a ____ attraction towards the nucleus and the atomic radius ____.
a stronger, decreases
b stronger, increases
c weaker, increases
d weaker, decreases
To solve this, we recall the periodic trends: As we move left to right across a period, the atomic number (and thus proton number) increases. This increases the nuclear charge. The outer electrons are in the same energy level (so shielding from inner electrons doesn't increase much), so the attraction between outer electrons and the nucleus becomes stronger. A stronger nuclear attraction pulls the outer electrons closer, so the atomic radius decreases.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. Stronger, decreases