QUESTION IMAGE
Question
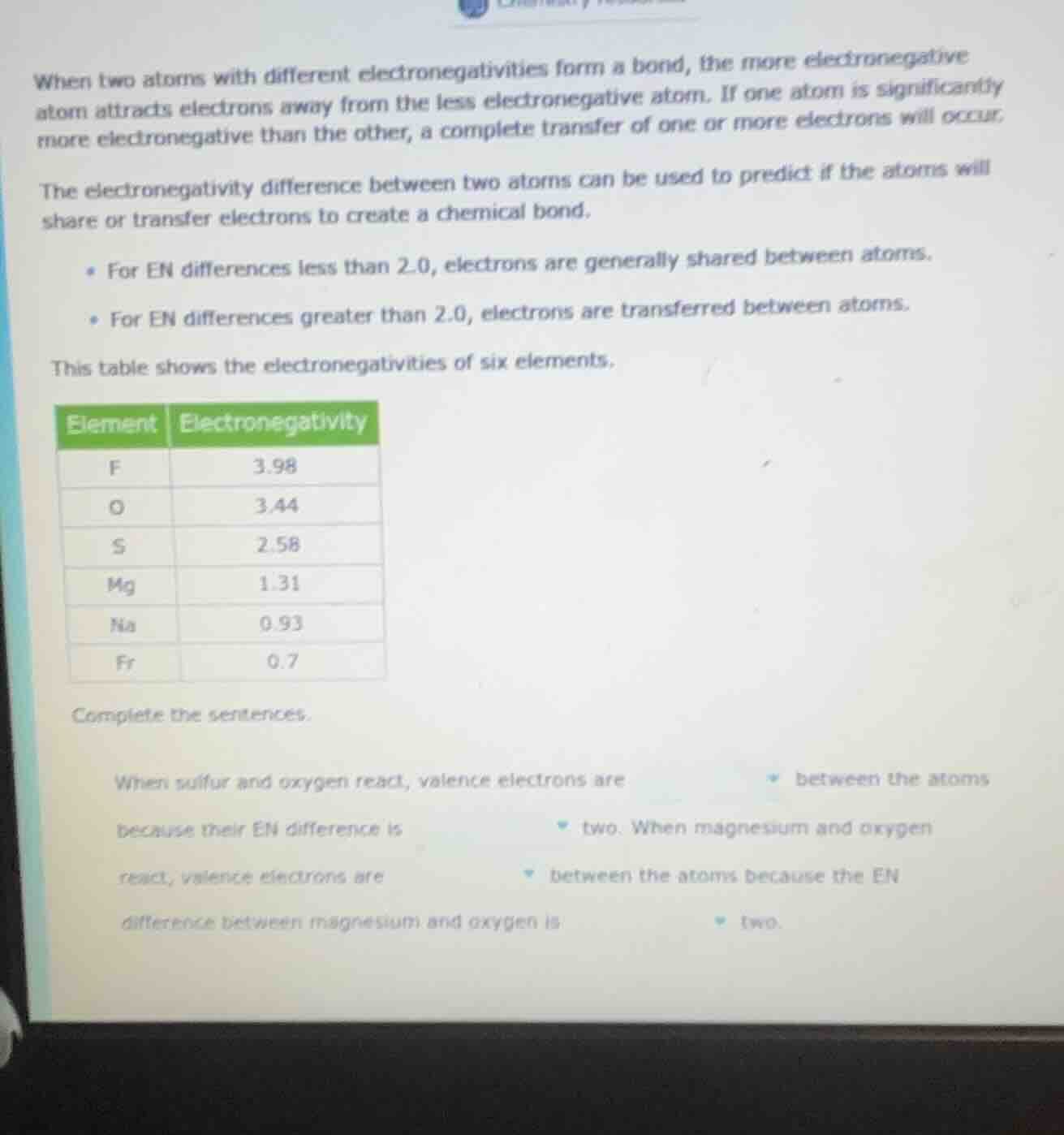
when two atoms with different electronegativities form a bond, the more electronegative atom attracts electrons away from the less electronegative atom. if one atom is significantly more electronegative than the other, a complete transfer of one or more electrons will occur.the electronegativity difference between two atoms can be used to predict if the atoms will share or transfer electrons to create a chemical bond.• for en differences less than 2.0, electrons are generally shared between atoms.• for en differences greater than 2.0, electrons are transferred between atoms.this table shows the electronegativities of six elements.element | electronegativityf | 3.98o | 3.44s | 2.58mg | 1.31na | 0.93fr | 0.7complete the sentences.when sulfur and oxygen react, valence electrons are between the atomsbecause their en difference is two. when magnesium and oxygenreact, valence electrons are between the atoms because the endifference between magnesium and oxygen is two.
Step1: Calculate S and O EN difference
$\Delta EN = 3.44 - 2.58 = 0.86$
Step2: Compare to 2.0, determine bond type
$0.86 < 2.0$, so electrons are shared.
Step3: Calculate Mg and O EN difference
$\Delta EN = 3.44 - 1.31 = 2.13$
Step4: Compare to 2.0, determine bond type
$2.13 > 2.0$, so electrons are transferred.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
When sulfur and oxygen react, valence electrons are shared between the atoms because their EN difference is less than two. When magnesium and oxygen react, valence electrons are transferred between the atoms because the EN difference between magnesium and oxygen is greater than two.