QUESTION IMAGE
Question
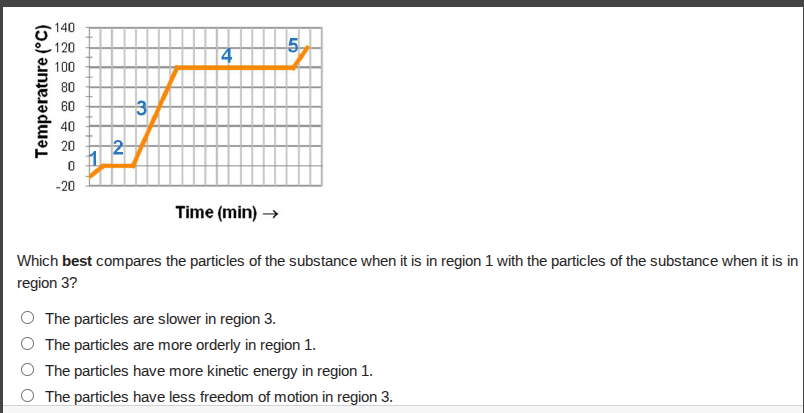
which best compares the particles of the substance when it is in region 1 with the particles of the substance when it is in region 3?
○ the particles are slower in region 3.
○ the particles are more orderly in region 1.
○ the particles have more kinetic energy in region 1.
○ the particles have less freedom of motion in region 3.
particles gain kinetic energy, while region 3's constant temp means kinetic energy stays the same. So particles in region 1 (as it warms) have more kinetic energy than the start of region 1, and compared to region 3, region 1's later particles have higher kinetic energy than region 3's particles (since region 3 temp is same as region 1's end of heating before phase change). Wait, no: Region 1: temp increases from -20 to 0, region 2: temp flat (melting), region 3: temp increases from 0 to 100, region 4: temp flat (boiling), region 5: temp increases from 100 to 120. So region 1 is warming a solid, region 3 is warming a liquid. Temperature is higher in region 3, but wait no: region 1 goes up to 0, region 3 starts at 0 and goes to 100. Wait the options: Option C says "The particles have more kinetic energy in region 1." No, wait no, region 3 has higher temperature, so higher kinetic energy. Wait no, I misread the graph. Wait region 1: temp starts at -20, rises to 0. Region 2: temp stays 0 (melting). Region 3: temp rises from 0 to 100. Region 4: temp stays 100 (boiling). Region 5: temp rises from 100 to 120. So region 3 has higher temperature than region 1, so particles in region 3 have higher kinetic energy. Wait the options:
A. The particles are slower in region 3. (False, region 3 is hotter, faster particles)
B. The particles are more orderly in region 1. (Region 1 is solid, region 3 is liquid: solid particles are more orderly, so this would be true? Wait no, region 1 is warming a solid, region 3 is warming a liquid. Solid particles are more orderly than liquid. But option B says "more orderly in region 1" which is true? Wait no, wait the question is "compares the particles of the substance when it is in region 1 with the particles when it is in region 3". Region 1 is solid (before melting), region 3 is liquid (after melting, warming). So solid particles (region1) are more orderly than liquid (region3). But option C says "more kinetic energy in region1" which is false, region3 is hotter, higher KE. Wait wait I messed up the graph. Wait region 3's temp goes up to 100, which is higher than region1's 0. So KE is higher in region3. So option A is false, B: more orderly in region1 (solid vs liquid: yes, solid is more orderly). Wait but option D: "less freedom of motion in region3" (liquid has more freedom than solid, so region3 (liquid) has more freedom, so D is false. Wait now I'm confused. Wait no, wait the graph: region 1: temp increases from -20 to 0 (solid warming), region2: temp 0 (melting, solid to liquid), region3: temp increases from 0 to 100 (liquid warming), region4: temp 100 (boiling, liquid to gas), region5: temp increases from 100 to 120 (gas warming). So:
A. Particles slower in region3: false, region3 is hotter, faster particles.
B. Particles more orderly in region1: true, solid (region1) is more orderly than liquid (region3).
C. More KE in region1: false, region3 is hotter, higher KE.
D. Less freedom in region3: false, liquid (region3) has more freedom than solid (region1).
Wait but that contradicts my earlier thought. Wait why did I think C was correct? I misread the graph. Wait no, wait the question says "when it is in region 1" vs "when it is in region3". Region1 is solid, region3 is liquid. So solid particles are more orderly (B is correct). But wait the option B says "The particles are more orderly in region 1." That is true. But wait let's check again. Wait maybe I misread the regions. Wait the labels: 1 is first segment (temp up), 2 is flat, 3 is temp up, 4 is flat,5 is temp up. Yes. So region1: solid, region3: liq…
Region 1 corresponds to a warming solid state, where particles are arranged in a fixed, orderly structure. Region 3 corresponds to a warming liquid state, where particles have more freedom to move and are less ordered. Temperature (and thus kinetic energy) is higher in region 3, so particles are faster there, eliminating options A, C, and D. Only option B accurately compares the particle order between the two regions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The particles are more orderly in region 1.