QUESTION IMAGE
Question
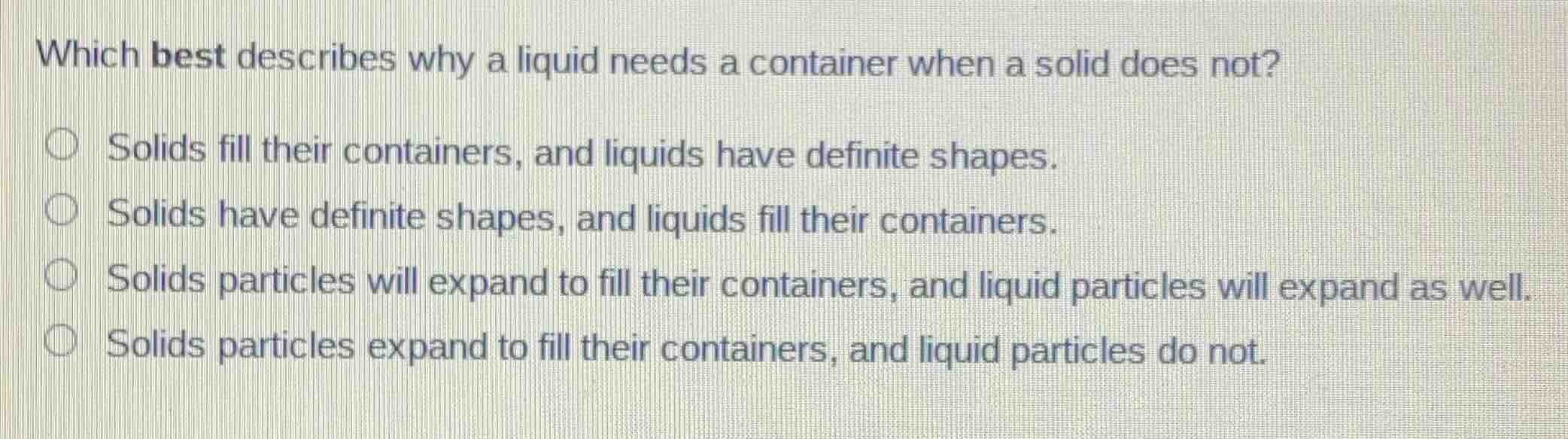
which best describes why a liquid needs a container when a solid does not?
○ solids fill their containers, and liquids have definite shapes.
○ solids have definite shapes, and liquids fill their containers.
○ solids particles will expand to fill their containers, and liquid particles will expand as well.
○ solids particles expand to fill their containers, and liquid particles do not.
Brief Explanations
To solve this, we analyze the properties of solids and liquids:
- Solids have a definite shape because their particles are closely packed and vibrate in fixed positions, so they don't need a container to maintain their shape.
- Liquids have particles that are close but can move past each other, so they take the shape of their container (fill the container's shape) and thus need a container to hold them.
- Analyzing the options:
- Option 1 is incorrect: Solids don't fill containers (liquids do), and liquids do not have definite shapes.
- Option 2 is correct: Solids have definite shapes (so no container needed for shape), and liquids fill their containers (so need a container).
- Option 3 is incorrect: Solid particles do not expand to fill containers; that's a gas property.
- Option 4 is incorrect: Solid particles don't expand to fill containers, and liquid particles do take the container's shape (fill it), so this is wrong.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Solids have definite shapes, and liquids fill their containers.