QUESTION IMAGE
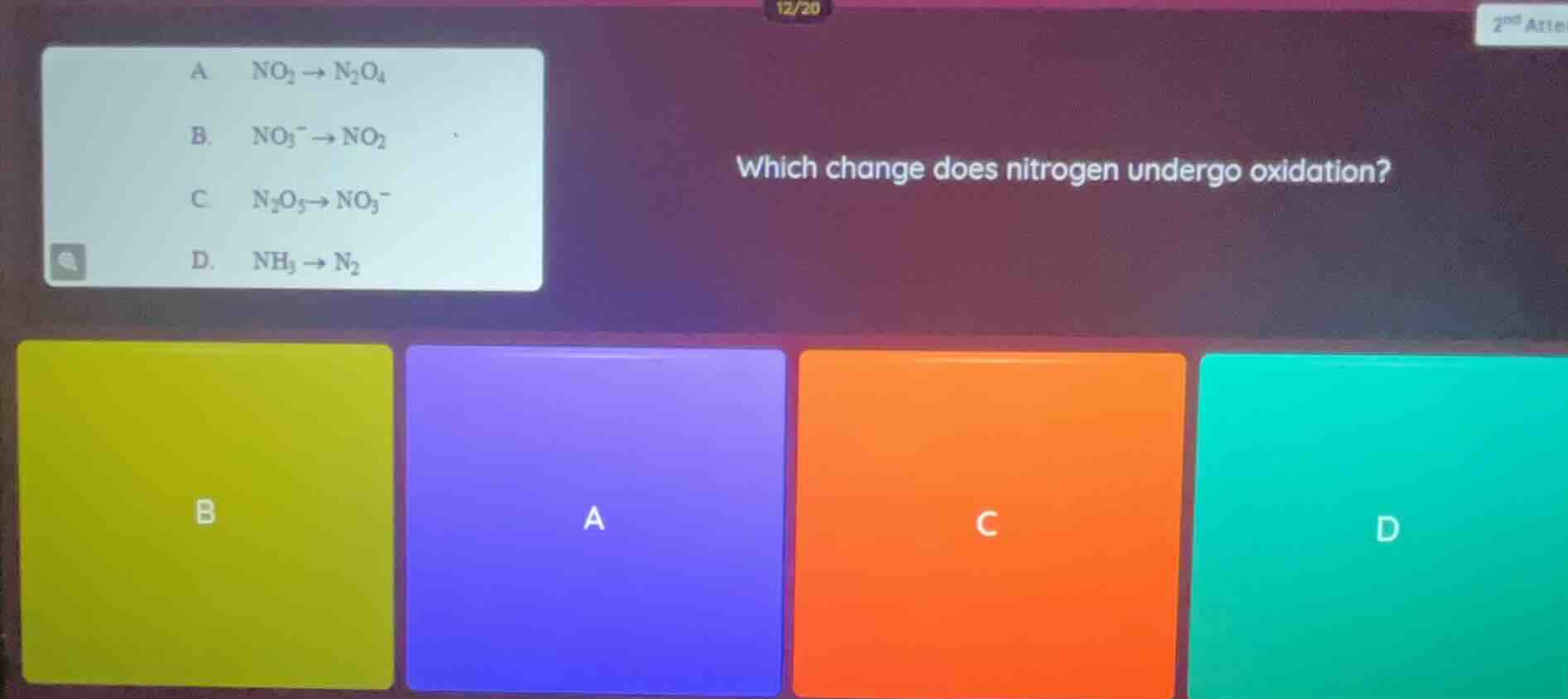
Question
which change does nitrogen undergo oxidation?
a. $ce{no_{2} -> n_{2}o_{4}}$
b. $ce{no_{3}^{-} -> no_{2}}$
c. $ce{n_{2}o_{5} -> no_{3}^{-}}$
d. $ce{nh_{3} -> n_{2}}$
Step1: Define oxidation state rule
Oxidation state of O = -2, H = +1; neutral compound: sum=0; ion: sum=charge.
Step2: Calculate A's N oxidation states
For $\text{NO}_2$: $x + 2(-2)=0 \implies x=+4$; For $\text{N}_2\text{O}_4$: $2x + 4(-2)=0 \implies x=+4$. No change.
Step3: Calculate B's N oxidation states
For $\text{NO}_3^-$: $x + 3(-2)=-1 \implies x=+5$; For $\text{NO}_2$: $x + 2(-2)=0 \implies x=+4$. Decrease (reduction).
Step4: Calculate C's N oxidation states
For $\text{N}_2\text{O}_5$: $2x + 5(-2)=0 \implies x=+5$; For $\text{NO}_3^-$: $x + 3(-2)=-1 \implies x=+5$. No change.
Step5: Calculate D's N oxidation states
For $\text{NH}_3$: $x + 3(+1)=0 \implies x=-3$; For $\text{N}_2$: $x=0$. Increase (oxidation).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. $\text{NH}_3
ightarrow \text{N}_2$