QUESTION IMAGE
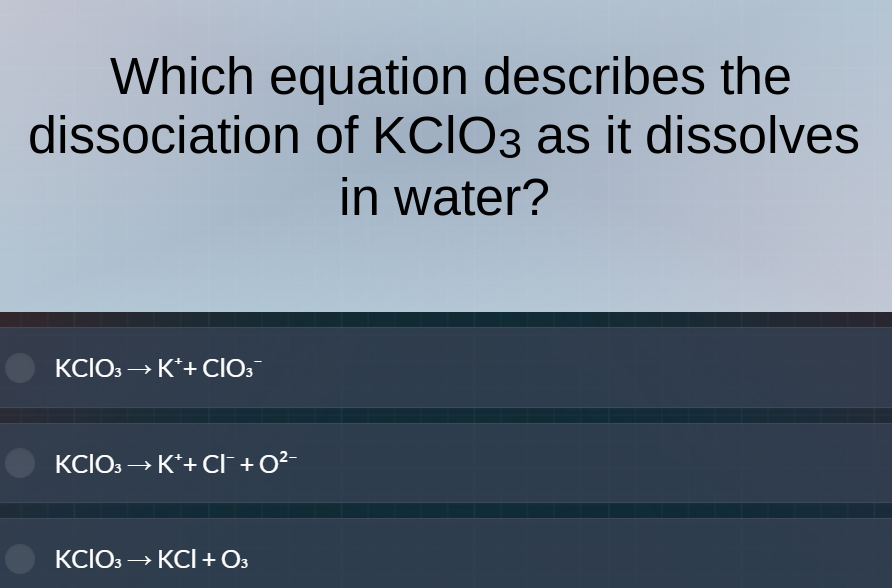
Question
which equation describes the dissociation of kclo₃ as it dissolves in water? kclo₃→k⁺+clo₃⁻ kclo₃→k⁺+cl⁻+o²⁻ kclo₃→kcl+o₃
To determine the correct dissociation equation of \( \text{KClO}_3 \) in water, we analyze the ionic composition. \( \text{KClO}_3 \) is a potassium chlorate, which is an ionic compound. When it dissolves in water, it dissociates into its constituent ions. The chlorate ion is \( \text{ClO}_3^- \), so \( \text{KClO}_3 \) should dissociate into \( \text{K}^+ \) (potassium ion) and \( \text{ClO}_3^- \) (chlorate ion). The second option incorrectly breaks down the chlorate ion into \( \text{Cl}^- \) and \( \text{O}^{2-} \), which is not how polyatomic ions dissociate in solution. The third option shows a decomposition reaction (forming \( \text{KCl} \) and \( \text{O}_3 \)) rather than a dissociation into ions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( \text{KClO}_3
ightarrow \text{K}^+ + \text{ClO}_3^- \)