QUESTION IMAGE
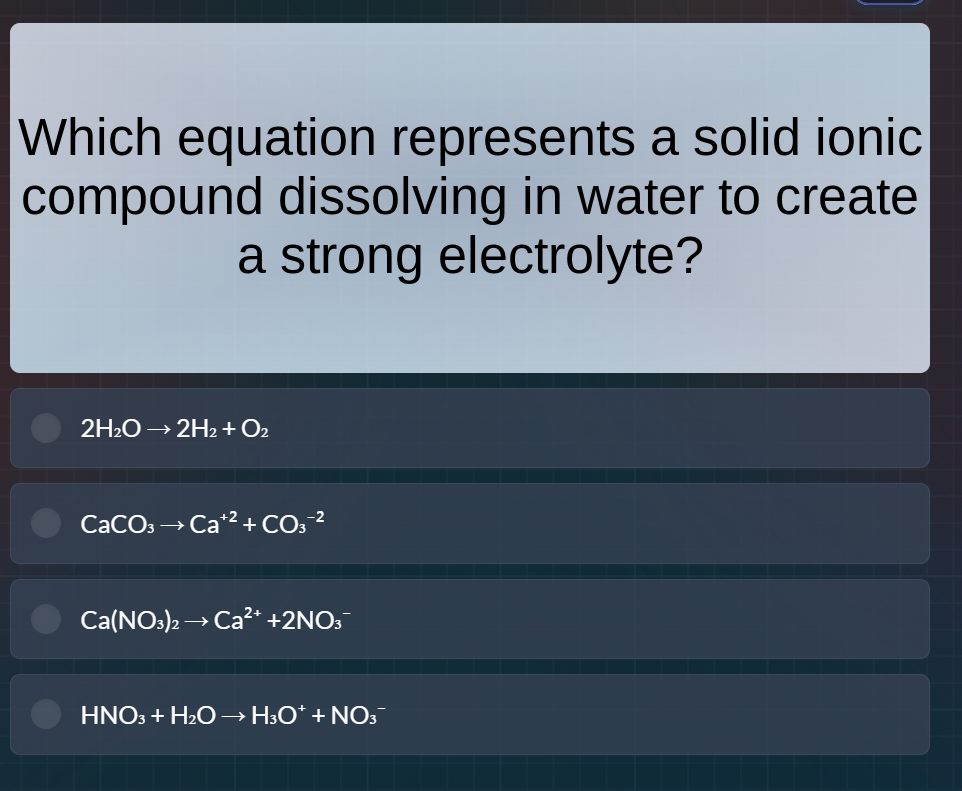
Question
which equation represents a solid ionic compound dissolving in water to create a strong electrolyte? 2h₂o → 2h₂ + o₂ caco₃ → ca⁺² + co₃⁻² ca(no₃)₂ → ca²⁺ + 2no₃⁻ hno₃ + h₂o → h₃o⁺ + no₃⁻
Brief Explanations
- Analyze the first option: \(2H_2O
ightarrow 2H_2 + O_2\) is the decomposition of water, not dissolution of an ionic compound.
- Analyze the second option: \(CaCO_3\) is a sparingly soluble salt, it does not dissociate completely in water, so it is not a strong electrolyte in terms of complete dissolution.
- Analyze the third option: \(Ca(NO_3)_2\) is a solid ionic compound. When it dissolves in water, it dissociates completely into \(Ca^{2 +}\) and \(2NO_3^-\) ions. Since it dissociates completely, it is a strong electrolyte.
- Analyze the fourth option: \(HNO_3\) is an acid, not a solid ionic compound. It reacts with water, but the reactant is not a solid ionic compound.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. \(Ca(NO_3)_2
ightarrow Ca^{2+} + 2NO_3^-\)