QUESTION IMAGE
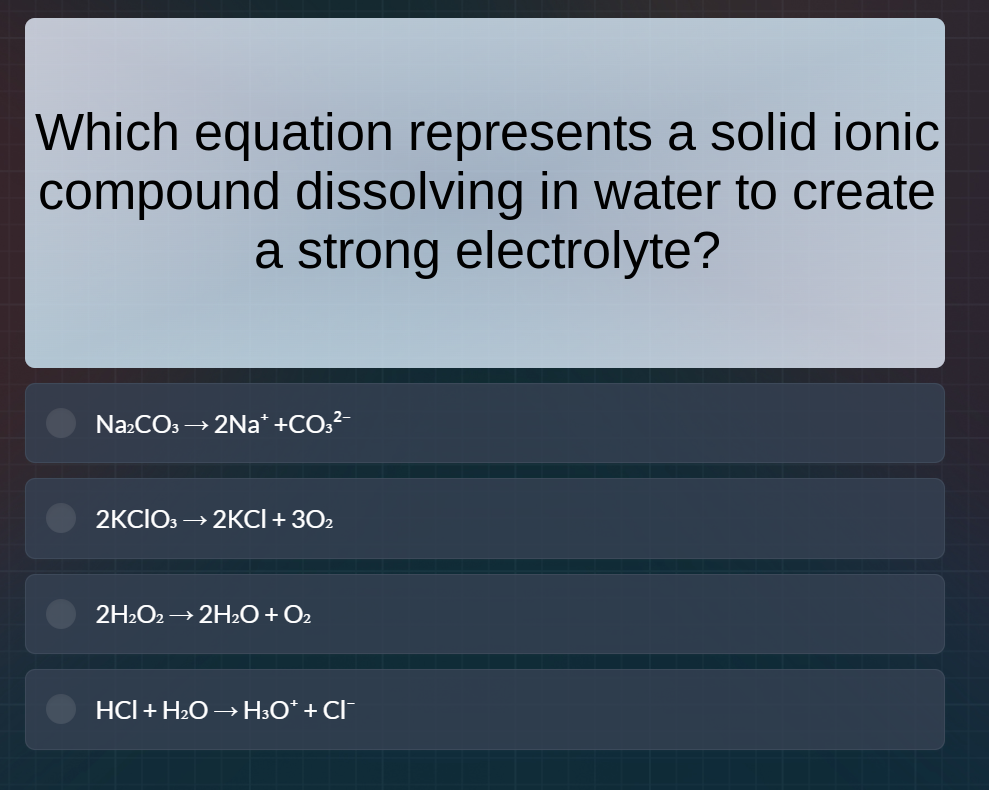
Question
which equation represents a solid ionic compound dissolving in water to create a strong electrolyte? na₂co₃ → 2na⁺ +co₃²⁻ 2kclo₃ → 2kcl + 3o₂ 2h₂o₂ → 2h₂o + o₂ hcl + h₂o → h₃o⁺ + cl⁻
Brief Explanations
To determine the correct equation, we analyze each option:
- For $\ce{Na_{2}CO_{3} -> 2Na^{+} + CO_{3}^{2-}}$: Sodium carbonate ($\ce{Na_{2}CO_{3}}$) is a solid ionic compound. When it dissolves in water, it dissociates into $\ce{Na^{+}}$ and $\ce{CO_{3}^{2-}}$ ions, and these ions make the solution a strong electrolyte (since strong electrolytes dissociate completely into ions in solution).
- For $\ce{2KClO_{3} -> 2KCl + 3O_{2}}$: This is a decomposition reaction of potassium chlorate to form potassium chloride and oxygen gas. It is not a dissolution of a solid ionic compound in water to form a strong electrolyte (it's a thermal decomposition reaction).
- For $\ce{2H_{2}O_{2} -> 2H_{2}O + O_{2}}$: This is the decomposition of hydrogen peroxide into water and oxygen. It is not related to the dissolution of a solid ionic compound in water.
- For $\ce{HCl + H_{2}O -> H_{3}O^{+} + Cl^{-}}$: Hydrochloric acid ($\ce{HCl}$) is a covalent compound (not a solid ionic compound in its pure state; $\ce{HCl}$ is a gas that dissolves in water) and this represents the ionization of $\ce{HCl}$ in water, but $\ce{HCl}$ is not a solid ionic compound.
So the equation $\ce{Na_{2}CO_{3} -> 2Na^{+} + CO_{3}^{2-}}$ represents a solid ionic compound (sodium carbonate) dissolving in water to create a strong electrolyte (since the ions are fully dissociated).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\ce{Na_{2}CO_{3} -> 2Na^{+} + CO_{3}^{2-}}$