QUESTION IMAGE
Question
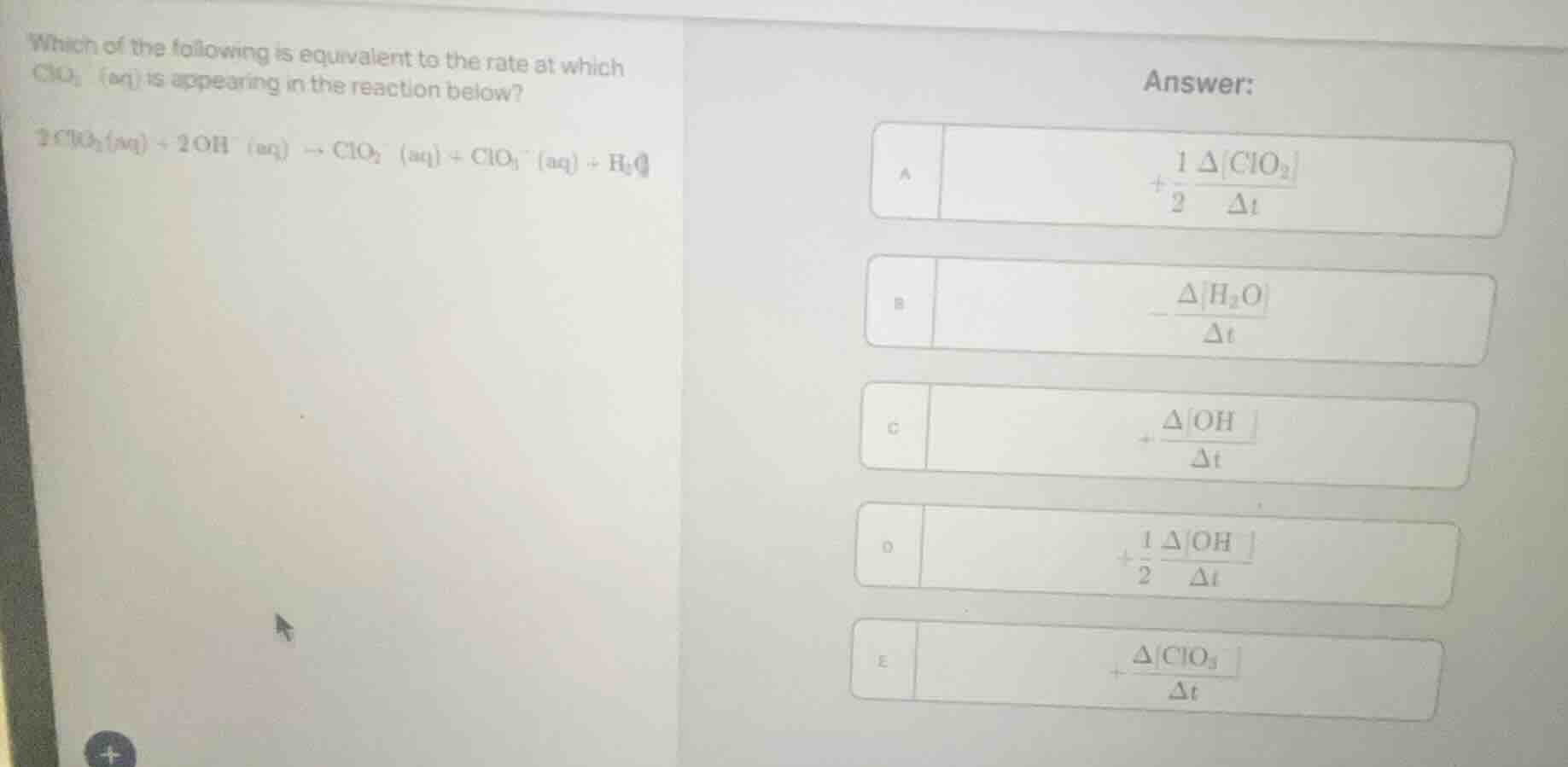
which of the following is equivalent to the rate at which clo₃⁻(aq) is appearing in the reaction below?
$2clo_{2}(aq)+2oh^{-}(aq)\
ightarrow clo_{2}^{-}(aq)+clo_{3}^{-}(aq)+h_{2}o(l)$
answer:
a. $+\frac{1}{2}\frac{\deltaclo_{2}}{\delta t}$
b. $-\frac{\deltah_{2}o}{\delta t}$
c. $+\frac{\deltaoh^{-}}{\delta t}$
d. $+\frac{1}{2}\frac{\deltaoh^{-}}{\delta t}$
e. $+\frac{\deltaclo_{3}^{-}}{\delta t}$
Step1: Recall reaction rate relationships
For a reaction $aA + bB
ightarrow cC + dD$, the rate is defined as:
$$-\frac{1}{a}\frac{\Delta[A]}{\Delta t} = -\frac{1}{b}\frac{\Delta[B]}{\Delta t} = \frac{1}{c}\frac{\Delta[C]}{\Delta t} = \frac{1}{d}\frac{\Delta[D]}{\Delta t}$$
Negative signs apply to reactants (concentration decreases over time).
Step2: Match to given reaction
The reaction is $2\text{ClO}_2(aq) + 2\text{OH}^-(aq)
ightarrow \text{ClO}_2^-(aq) + \text{ClO}_3^-(aq) + \text{H}_2\text{O}(l)$
We need the rate for $\text{ClO}_3^-$ (product, positive sign, stoichiometric coefficient 1):
$$\text{Rate} = \frac{\Delta[\text{ClO}_3^-]}{\Delta t}$$
Compare to each option:
- Option A: $\frac{1}{2}\frac{\Delta[\text{ClO}_2^-]}{\Delta t}$: $\text{ClO}_2^-$ has coefficient 1, so $\frac{\Delta[\text{ClO}_2^-]}{\Delta t} = \frac{\Delta[\text{ClO}_3^-]}{\Delta t}$, so this is not equivalent.
- Option B: $-\frac{\Delta[\text{H}_2\text{O}]}{\Delta t}$: $\text{H}_2\text{O}$ is a product, so its rate should be positive, and its coefficient is 1, so $\frac{\Delta[\text{H}_2\text{O}]}{\Delta t} = \frac{\Delta[\text{ClO}_3^-]}{\Delta t}$, the negative sign makes this incorrect.
- Option C: $\frac{\Delta[\text{OH}^-]}{\Delta t}$: $\text{OH}^-$ is a reactant, so its rate should be negative, and $\text{Rate} = -\frac{1}{2}\frac{\Delta[\text{OH}^-]}{\Delta t} = \frac{\Delta[\text{ClO}_3^-]}{\Delta t}$, so this is not equivalent.
- Option D: $\frac{1}{2}\frac{\Delta[\text{OH}^-]}{\Delta t}$: $\text{OH}^-$ is a reactant, so $\text{Rate} = -\frac{1}{2}\frac{\Delta[\text{OH}^-]}{\Delta t} = \frac{\Delta[\text{ClO}_3^-]}{\Delta t}$, the positive sign makes this incorrect.
- Option E: $\frac{\Delta[\text{ClO}_3^-]}{\Delta t}$: This directly matches the rate of appearance of $\text{ClO}_3^-$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
E. $+\frac{\Delta[\text{ClO}_3^-]}{\Delta t}$