QUESTION IMAGE
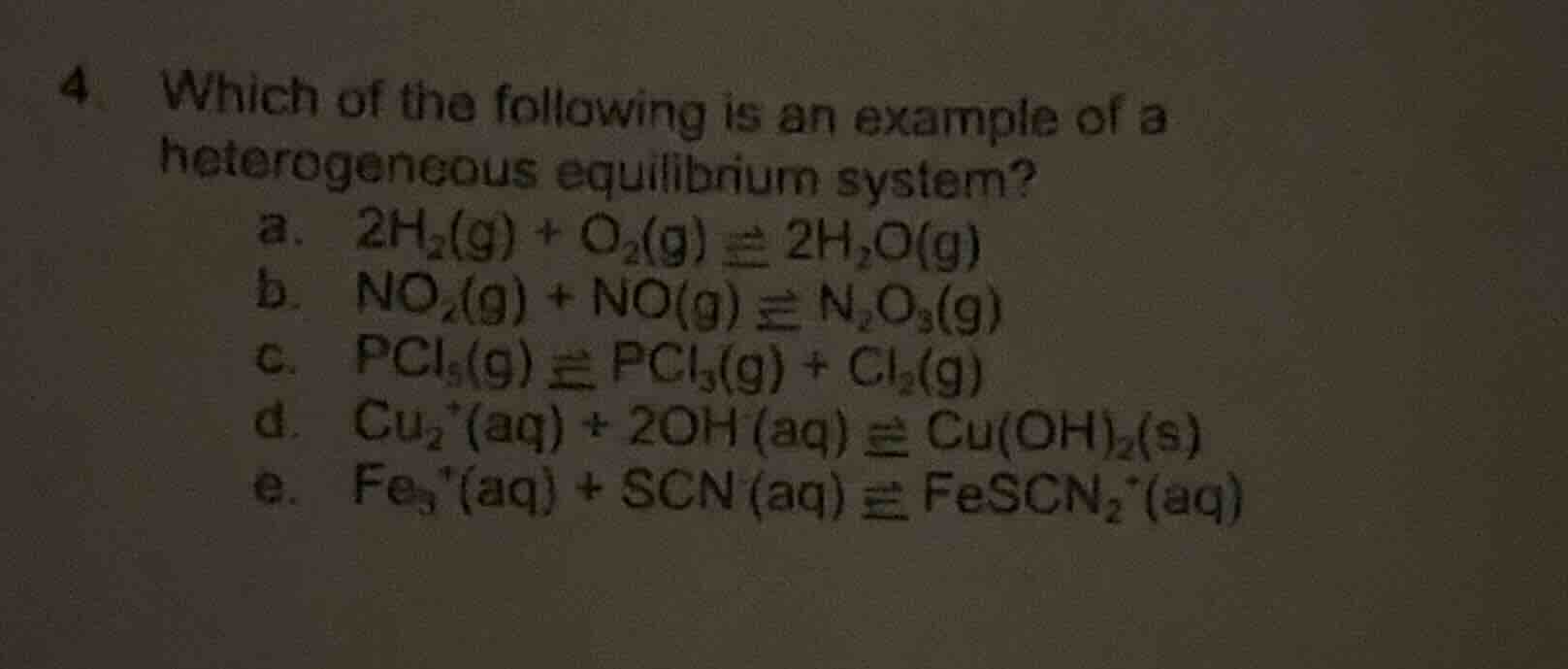
Question
4 which of the following is an example of a heterogeneous equilibrium system?
a. $2\ce{h_{2}(g) + o_{2}(g) \
ightleftharpoons 2h_{2}o(g)}$
b. $\ce{no_{2}(g) + no(g) \
ightleftharpoons n_{2}o_{3}(g)}$
c. $\ce{pcl_{5}(g) \
ightleftharpoons pcl_{3}(g) + cl_{2}(g)}$
d. $\ce{cu^{2+}(aq) + 2oh^{-}(aq) \
ightleftharpoons cu(oh)_{2}(s)}$
e. $\ce{fe^{3+}(aq) + scn^{-}(aq) \
ightleftharpoons fescn^{2+}(aq)}$
A heterogeneous equilibrium involves reactants and products in different phases (states of matter). Let's analyze each option:
- Option a: All species are gaseous (g), so homogeneous.
- Option b: All species are gaseous (g), homogeneous.
- Option c: All species are gaseous (g), homogeneous.
- Option d: Reactants are aqueous (aq), product is solid (s) – different phases, so heterogeneous.
- Option e: All species are aqueous (aq), homogeneous.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
d. $\ce{Cu^{2+}(aq) + 2OH^{-}(aq)
ightleftharpoons Cu(OH)_{2}(s)}$