QUESTION IMAGE
Question
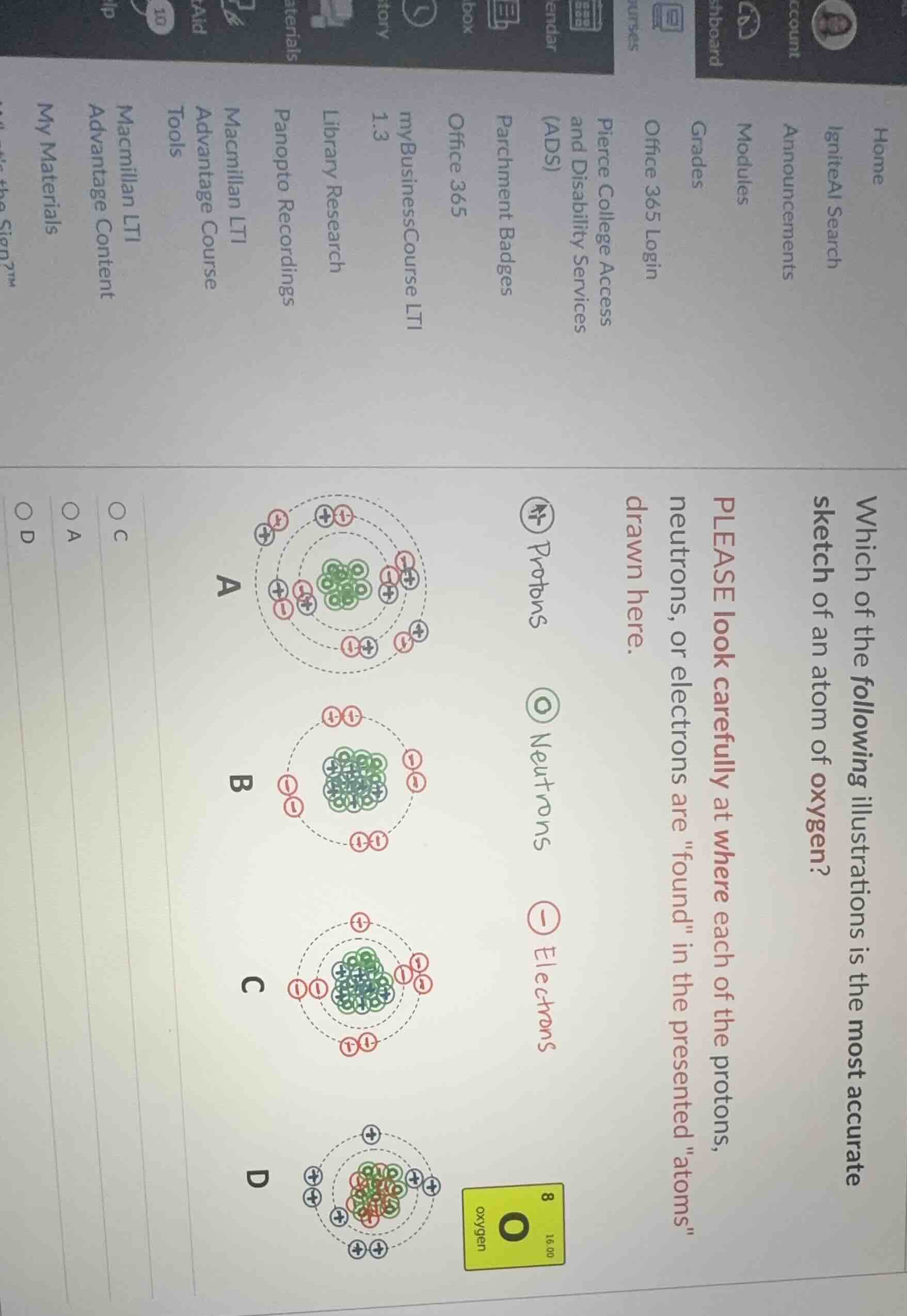
which of the following illustrations is the most accurate sketch of an atom of oxygen?
please look carefully at where each of the protons, neutrons, or electrons are \found\ in the presented \atoms\ drawn here.
protons: ⊕, neutrons: ◎, electrons: ⊖
options: a, b, c, d
(with oxygen element information: o, atomic number 8, atomic mass 16.00)
Step1: Identify oxygen's subatomic counts
Oxygen (O) has an atomic number of 8, so it has 8 protons (\(p^+=8\)) and 8 electrons (\(e^-=8\)) in a neutral atom. Its mass number is ~16, so neutrons (\(n^0=16-8=8\)).
Step2: Check subatomic locations
Protons + neutrons are in the nucleus; electrons orbit the nucleus.
Step3: Evaluate each option
- A: Protons are outside the nucleus (incorrect).
- B: Has 8 electrons, but protons are outside the nucleus (incorrect).
- C: 8 protons/neutrons in nucleus, 8 electrons in orbitals (matches oxygen's properties).
- D: No electrons present (incorrect).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C