QUESTION IMAGE
Question
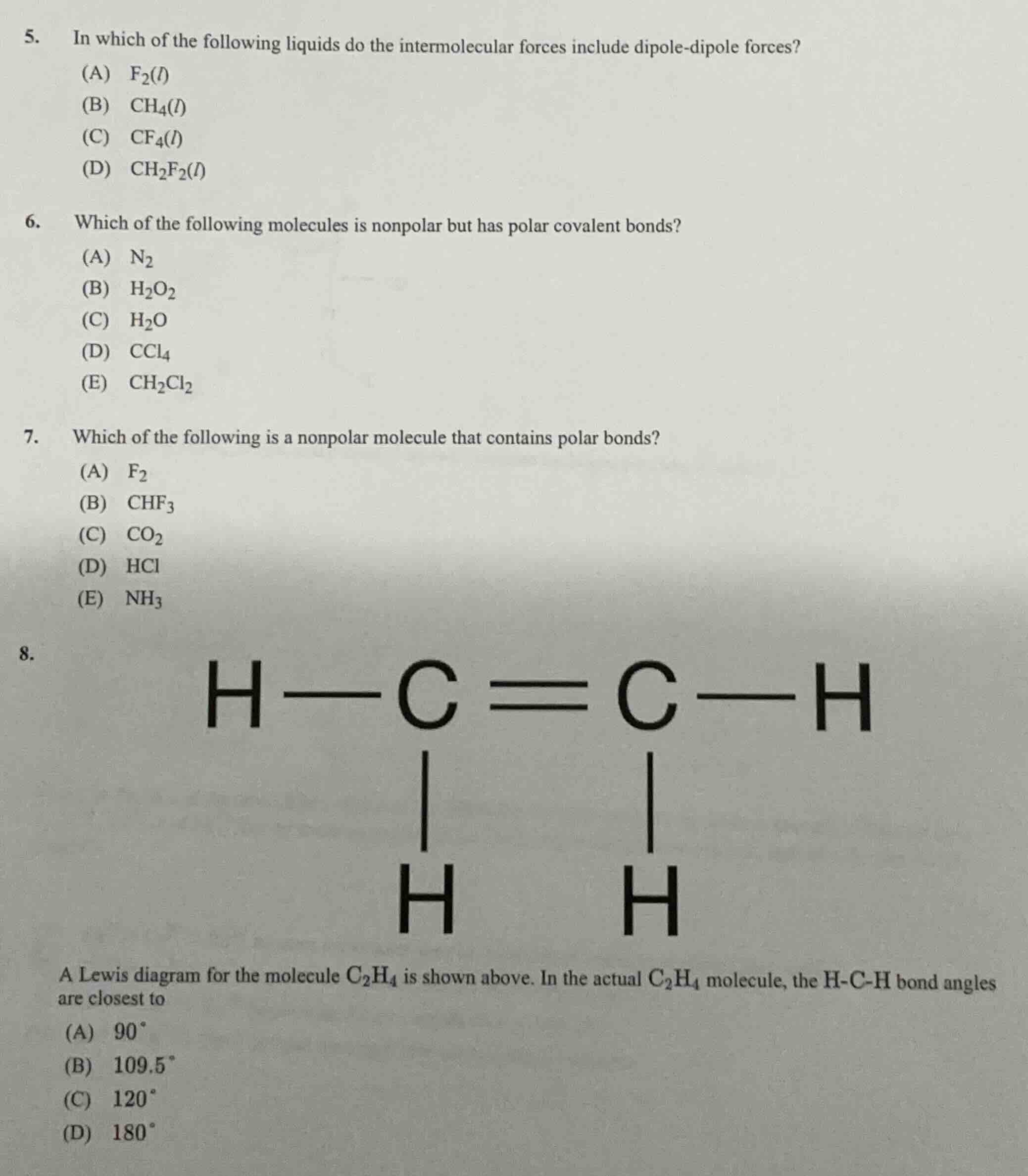
- in which of the following liquids do the intermolecular forces include dipole-dipole forces?(a) $ce{f_{2}(l)}$(b) $ce{ch_{4}(l)}$(c) $ce{cf_{4}(l)}$(d) $ce{ch_{2}f_{2}(l)}$6. which of the following molecules is nonpolar but has polar covalent bonds?(a) $ce{n_{2}}$(b) $ce{h_{2}o_{2}}$(c) $ce{h_{2}o}$(d) $ce{ccl_{4}}$(e) $ce{ch_{2}cl_{2}}$7. which of the following is a nonpolar molecule that contains polar bonds?(a) $ce{f_{2}}$(b) $ce{chf_{3}}$(c) $ce{co_{2}}$(d) $ce{hcl}$(e) $ce{nh_{3}}$8. a lewis diagram for the molecule $ce{c_{2}h_{4}}$ is shown above. in the actual $ce{c_{2}h_{4}}$ molecule, the h-c-h bond angles are closest to(a) $90^circ$(b) $109.5^circ$(c) $120^circ$(d) $180^circ$
Brief Explanations
Question 5:
Dipole-dipole forces exist in polar molecules. Nonpolar molecules (symmetric, equal bond polarities cancel out) only have London dispersion forces.
- (A) $\text{F}_2$: Same atoms, nonpolar.
- (B) $\text{CH}_4$: Tetrahedral, symmetric, nonpolar.
- (C) $\text{CF}_4$: Tetrahedral, symmetric, nonpolar.
- (D) $\text{CH}_2\text{F}_2$: Tetrahedral, unequal substituents, polar.
Question 6:
A nonpolar molecule with polar bonds has symmetric geometry that cancels bond dipoles.
- (A) $\text{N}_2$: Nonpolar, nonpolar bonds.
- (B) $\text{H}_2\text{O}_2$: Polar molecule, polar bonds.
- (C) $\text{H}_2\text{O}$: Polar molecule, polar bonds.
- (D) $\text{CCl}_4$: Tetrahedral, symmetric, nonpolar; $\text{C-Cl}$ are polar bonds.
- (E) $\text{CH}_2\text{Cl}_2$: Polar molecule, polar bonds.
Question 7:
A nonpolar molecule with polar bonds has symmetric geometry that cancels bond dipoles.
- (A) $\text{F}_2$: Nonpolar, nonpolar bonds.
- (B) $\text{CHF}_3$: Polar molecule, polar bonds.
- (C) $\text{CO}_2$: Linear, symmetric, nonpolar; $\text{C=O}$ are polar bonds.
- (D) $\text{HCl}$: Polar molecule, polar bond.
- (E) $\text{NH}_3$: Polar molecule, polar bonds.
Question 8:
$\text{C}_2\text{H}_4$ has each C atom with $sp^2$ hybridization (trigonal planar electron geometry), which has bond angles close to $120^\circ$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- (D) $\text{CH}_2\text{F}_2(l)$
- (D) $\text{CCl}_4$
- (C) $\text{CO}_2$
- (C) $120^\circ$