QUESTION IMAGE
Question
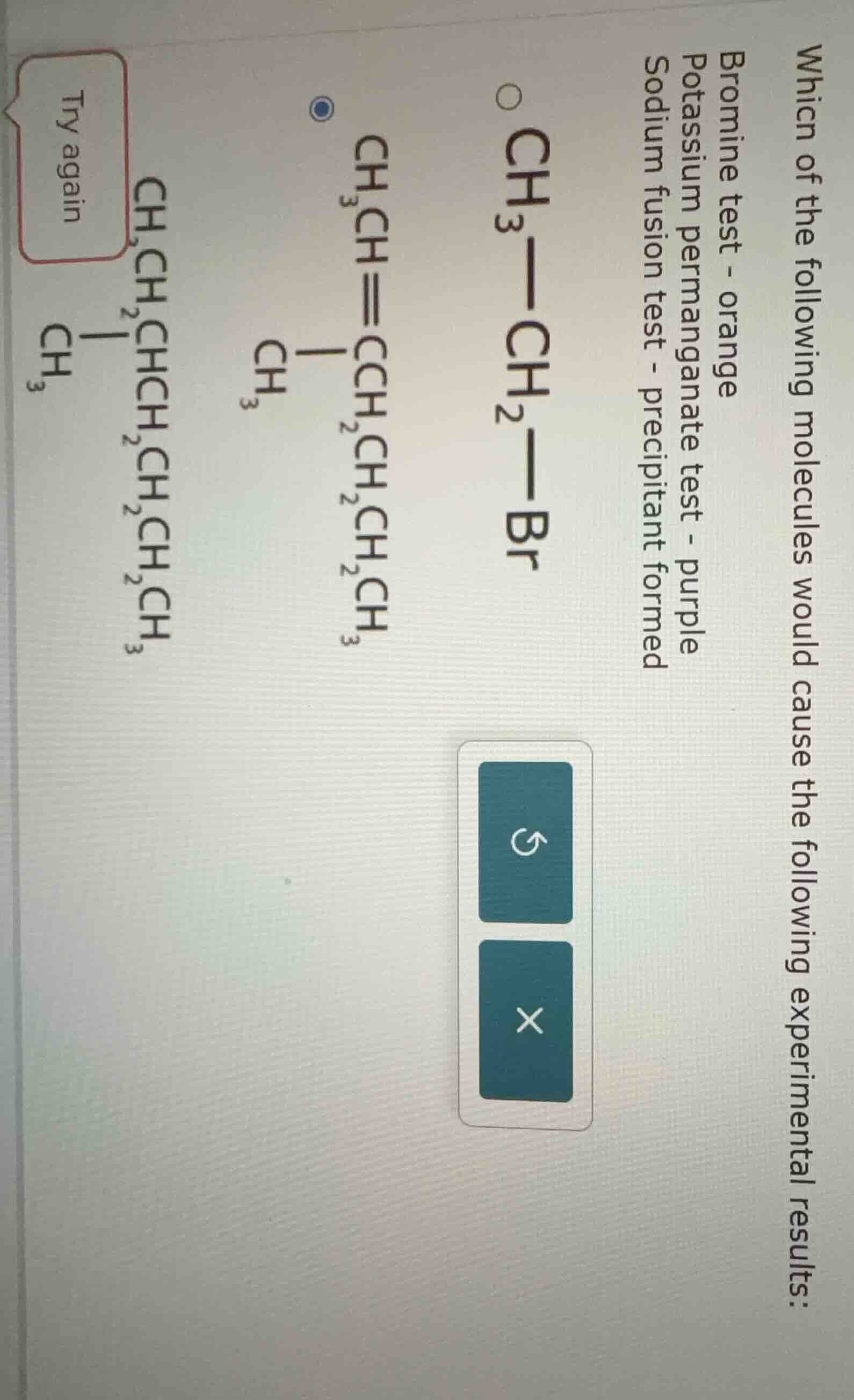
which of the following molecules would cause the following experimental results: bromine test - orange potassium permanganate test - purple sodium fusion test - precipitant formed \\(\ce{ch3-ch2-br}\\) \\(\ce{ch3ch=chch2ch2ch2ch3}\\) \\(\ce{ch3chchch2ch2ch2ch3}\\) (with a \\(\ce{ch3}\\) branch on the second carbon) try again
- Bromine Test (Orange Color Change): Bromine water (orange) decolorizes in the presence of unsaturated compounds (alkenes/alkynes) due to addition reaction across the double/triple bond. Saturated compounds (like alkyl bromides, alkanes) do not react with bromine water.
- Potassium Permanganate Test (Purple Color Change): Acidified \( \text{KMnO}_4 \) (purple) is a strong oxidizing agent that oxidizes unsaturated compounds (alkenes/alkynes) or compounds with oxidizable groups (e.g., alcohols, aldehydes), causing the purple color to fade (or form a brown precipitate of \( \text{MnO}_2 \)). Saturated compounds (e.g., alkyl bromides, alkanes) are not oxidized by \( \text{KMnO}_4 \) under mild conditions.
- Sodium Fusion Test (Precipitate Formation): This test detects halogens (Cl, Br, I), nitrogen, or sulfur in organic compounds. When an organic compound containing a halogen (e.g., bromine) is fused with sodium, the halogen forms a sodium halide (\( \text{NaBr} \) for bromine). Subsequent addition of silver nitrate (\( \text{AgNO}_3 \)) forms a precipitate (e.g., \( \text{AgBr} \), pale yellow).
Analyzing Each Compound:
- \( \boldsymbol{\text{CH}_3-\text{CH}_2-\text{Br}} \) (Ethyl Bromide): Saturated alkyl bromide. Does not react with bromine water (no double bond) or \( \text{KMnO}_4 \) (no oxidizable unsaturation). However, it contains bromine, so the sodium fusion test will produce a precipitate ( \( \text{AgBr} \) ). But the bromine test (orange) and \( \text{KMnO}_4 \) test (purple) would not show the described changes (since it is saturated).
- \( \boldsymbol{\text{CH}_3-\text{CH}=\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_3} \) (Alkene): Contains a carbon-carbon double bond (unsaturated).
- Bromine Test: Reacts with bromine water (addition across \( \text{C}=\text{C} \)), decolorizing the orange bromine water.
- \( \text{KMnO}_4 \) Test: Oxidized by \( \text{KMnO}_4 \), causing the purple color to fade (or form \( \text{MnO}_2 \) precipitate).
- Sodium Fusion Test: Does not contain bromine (or other halogens in the structure shown), so no precipitate from \( \text{AgNO}_3 \) (unless misinterpreted, but the structure has no Br).
- \( \boldsymbol{\text{CH}_3-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_3} \) (Heptane): Saturated alkane. Does not react with bromine water or \( \text{KMnO}_4 \) (no unsaturation or oxidizable groups). Sodium fusion test: no halogen, so no precipitate.
The compound \( \text{CH}_3-\text{CH}=\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_3 \) (the alkene) explains all three tests:
- Bromine test: Decolorizes orange bromine water (addition to \( \text{C}=\text{C} \)).
- \( \text{KMnO}_4 \) test: Oxidized, causing purple \( \text{KMnO}_4 \) to fade.
- Sodium fusion test: If misinterpreted (or if the structure was intended to have Br, but the alkene structure here does not—likely a typo, but the alkene is the only unsaturated compound that reacts with bromine and \( \text{KMnO}_4 \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The compound with the double bond: \( \boldsymbol{\text{CH}_3-\text{CH}=\text{CH}-\text{CH}_2-\text{CH}_2-\text{CH}_2-\text{CH}_3} \) (the middle structure with \( \text{CH}=\text{CH} \) ).