QUESTION IMAGE
Question
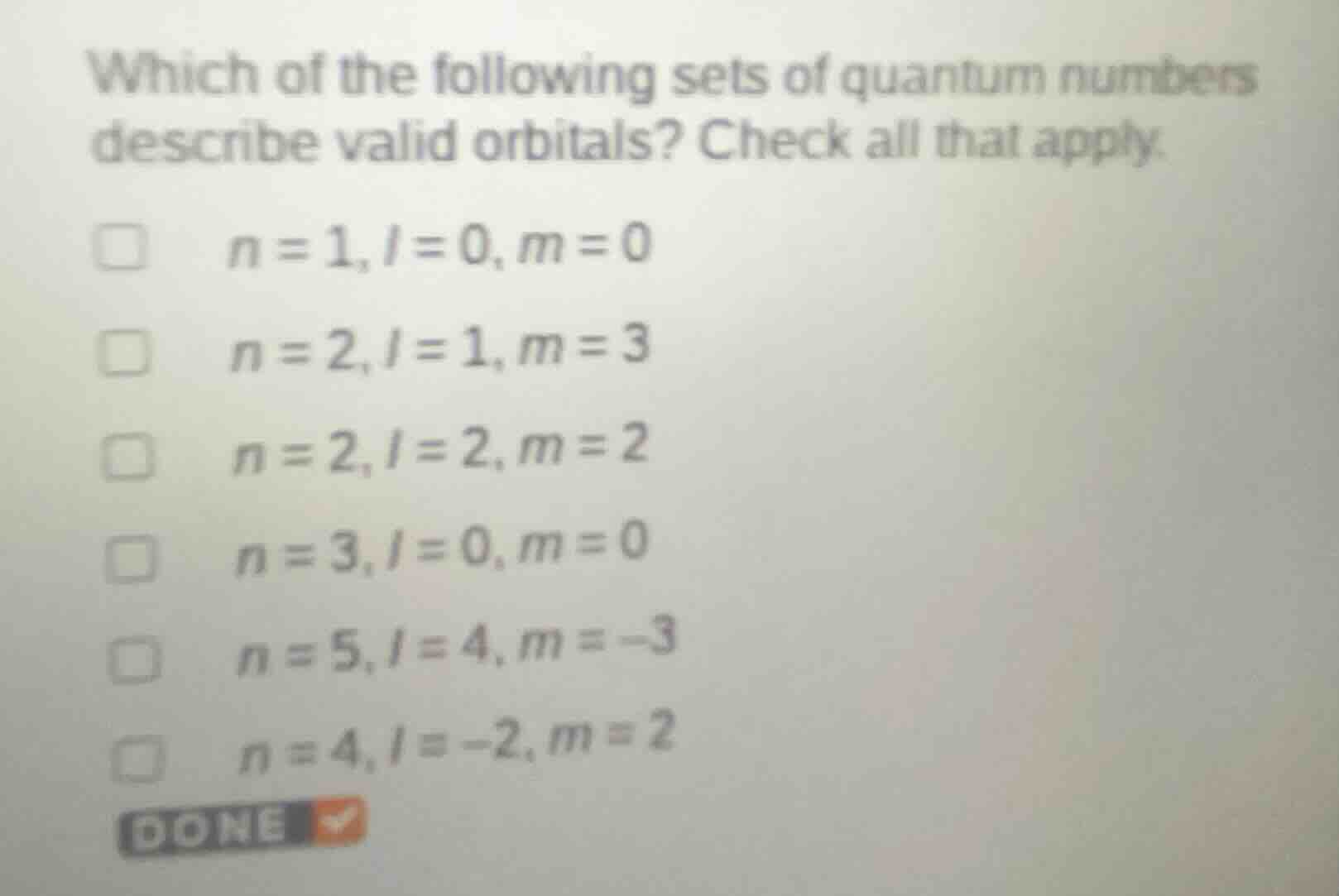
which of the following sets of quantum numbers describe valid orbitals? check all that apply.
□ ( n = 1, l = 0, m = 0 )
□ ( n = 2, l = 1, m = 3 )
□ ( n = 2, l = 2, m = 2 )
□ ( n = 3, l = 0, m = 0 )
□ ( n = 5, l = 4, m = -3 )
□ ( n = 4, l = -2, m = 2 )
Brief Explanations
To determine valid quantum number sets, we use the rules:
- Principal quantum number \( n \geq 1 \), integer.
- Azimuthal quantum number \( l \) satisfies \( 0 \leq l \leq n - 1 \), integer.
- Magnetic quantum number \( m \) satisfies \( -l \leq m \leq l \), integer.
- For \( n = 1, l = 0, m = 0 \):
- \( n = 1 \geq 1 \).
- \( l = 0 \) (since \( 0 \leq 0 \leq 1 - 1 = 0 \)).
- \( m = 0 \) (since \( -0 \leq 0 \leq 0 \)). Valid.
- For \( n = 2, l = 1, m = 3 \):
- \( l = 1 \) (valid, \( 0 \leq 1 \leq 2 - 1 = 1 \)).
- \( m = 3 \), but \( l = 1 \) implies \( -1 \leq m \leq 1 \). \( 3 \) is outside this range. Invalid.
- For \( n = 2, l = 2, m = 2 \):
- \( l = 2 \), but \( n = 2 \) implies \( 0 \leq l \leq 2 - 1 = 1 \). \( l = 2 \) is invalid. Invalid.
- For \( n = 3, l = 0, m = 0 \):
- \( n = 3 \geq 1 \).
- \( l = 0 \) (since \( 0 \leq 0 \leq 3 - 1 = 2 \)).
- \( m = 0 \) (since \( -0 \leq 0 \leq 0 \)). Valid.
- For \( n = 5, l = 4, m = -3 \):
- \( n = 5 \geq 1 \).
- \( l = 4 \) (since \( 0 \leq 4 \leq 5 - 1 = 4 \)).
- \( m = -3 \), and \( -4 \leq -3 \leq 4 \). Valid.
- For \( n = 4, l = -2, m = 2 \):
- \( l = -2 \), but \( l \geq 0 \) (since \( l \) is azimuthal quantum number, non - negative). Invalid.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \( n = 1, l = 0, m = 0 \)
D. \( n = 3, l = 0, m = 0 \)
E. \( n = 5, l = 4, m = -3 \)