QUESTION IMAGE
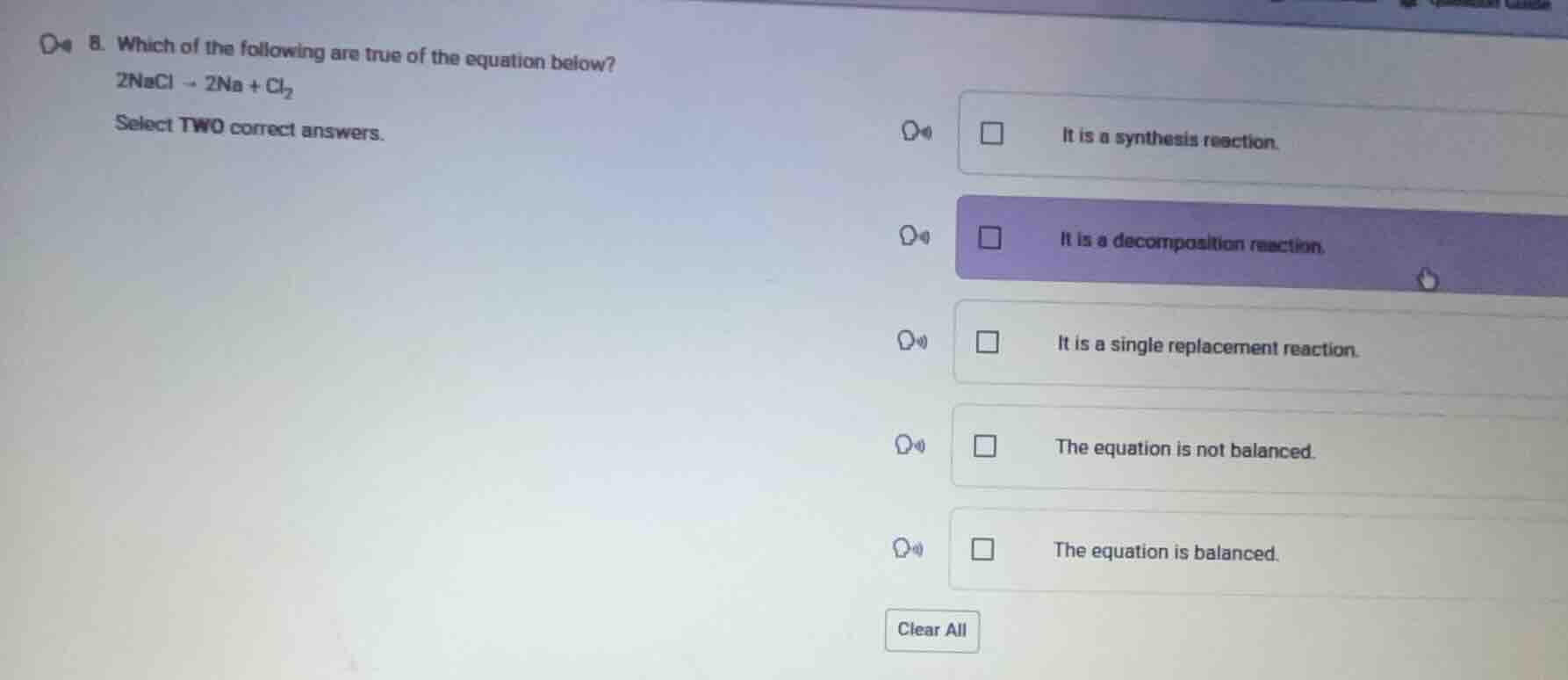
Question
- which of the following are true of the equation below?
$2\ce{nacl} \
ightarrow 2\ce{na} + \ce{cl_{2}}$
select two correct answers.
\\(\square\\) it is a synthesis reaction.
\\(\square\\) it is a decomposition reaction.
\\(\square\\) it is a single replacement reaction.
\\(\square\\) the equation is not balanced.
\\(\square\\) the equation is balanced.
clear all
Brief Explanations
- Reaction Type Analysis: A decomposition reaction is when a single compound breaks down into two or more simpler substances. Here, \(2\text{NaCl}\) (a single compound) breaks down into \(2\text{Na}\) and \(\text{Cl}_2\), so it is a decomposition reaction.
- Balancing the Equation: Count the atoms of each element on both sides. On the left: \(2\) Na, \(2\) Cl (from \(2\text{NaCl}\)). On the right: \(2\) Na (from \(2\text{Na}\)) and \(2\) Cl (from \(\text{Cl}_2\)). So the equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. It is a decomposition reaction
E. The equation is balanced