QUESTION IMAGE
Question
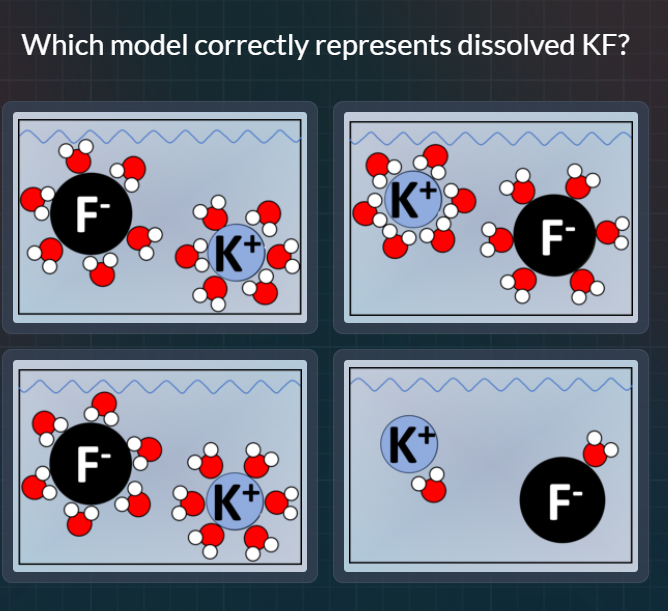
which model correctly represents dissolved kf?
When KF (potassium fluoride) dissolves in water, it dissociates into \( K^+ \) (potassium cation) and \( F^- \) (fluoride anion). Water molecules are polar, with the oxygen atom being partially negative and the hydrogen atoms being partially positive. For the \( K^+ \) ion (positive charge), the partially negative oxygen atoms of water molecules should orient towards it. For the \( F^- \) ion (negative charge), the partially positive hydrogen atoms of water molecules should orient towards it.
- In the first option: The orientation around \( K^+ \) and \( F^- \) does not follow the polar attraction (O towards \( K^+ \), H towards \( F^- \)) correctly.
- In the second option: The \( K^+ \) has water molecules with O (red) towards it (correct for positive ion), and \( F^- \) has H (white) towards it (correct for negative ion) as water's O is partial - and H is partial +.
- In the third option: The orientation around the ions does not match the polar attraction (incorrect arrangement of water molecules around the ions).
- In the fourth option: The ions are not properly solvated (water molecules around them do not follow the polar attraction rules).
So the second model (top - right) correctly represents dissolved KF.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The model in the top - right (the second model when looking at the four models: top - left, top - right, bottom - left, bottom - right) correctly represents dissolved KF. In this model, \( K^+ \) is surrounded by water molecules with the oxygen (red) part towards it (correct for a positive ion) and \( F^- \) is surrounded by water molecules with the hydrogen (white) part towards it (correct for a negative ion), following the polar nature of water and the charge - dipole interactions during solvation of KF.