QUESTION IMAGE
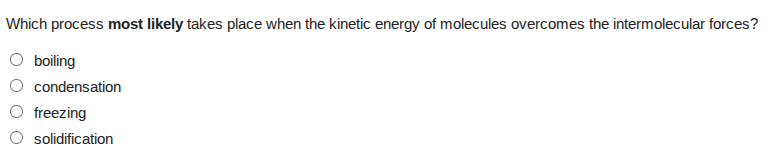
Question
which process most likely takes place when the kinetic energy of molecules overcomes the intermolecular forces?
boiling
condensation
freezing
solidification
, freezing, and solidification all involve a decrease in molecular kinetic energy, where intermolecular forces become stronger to form a more ordered state (liquid from gas, solid from liquid, respectively).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
, freezing, and solidification all involve a decrease in molecular kinetic energy, where intermolecular forces become stronger to form a more ordered state (liquid from gas, solid from liquid, respectively).