QUESTION IMAGE
Question
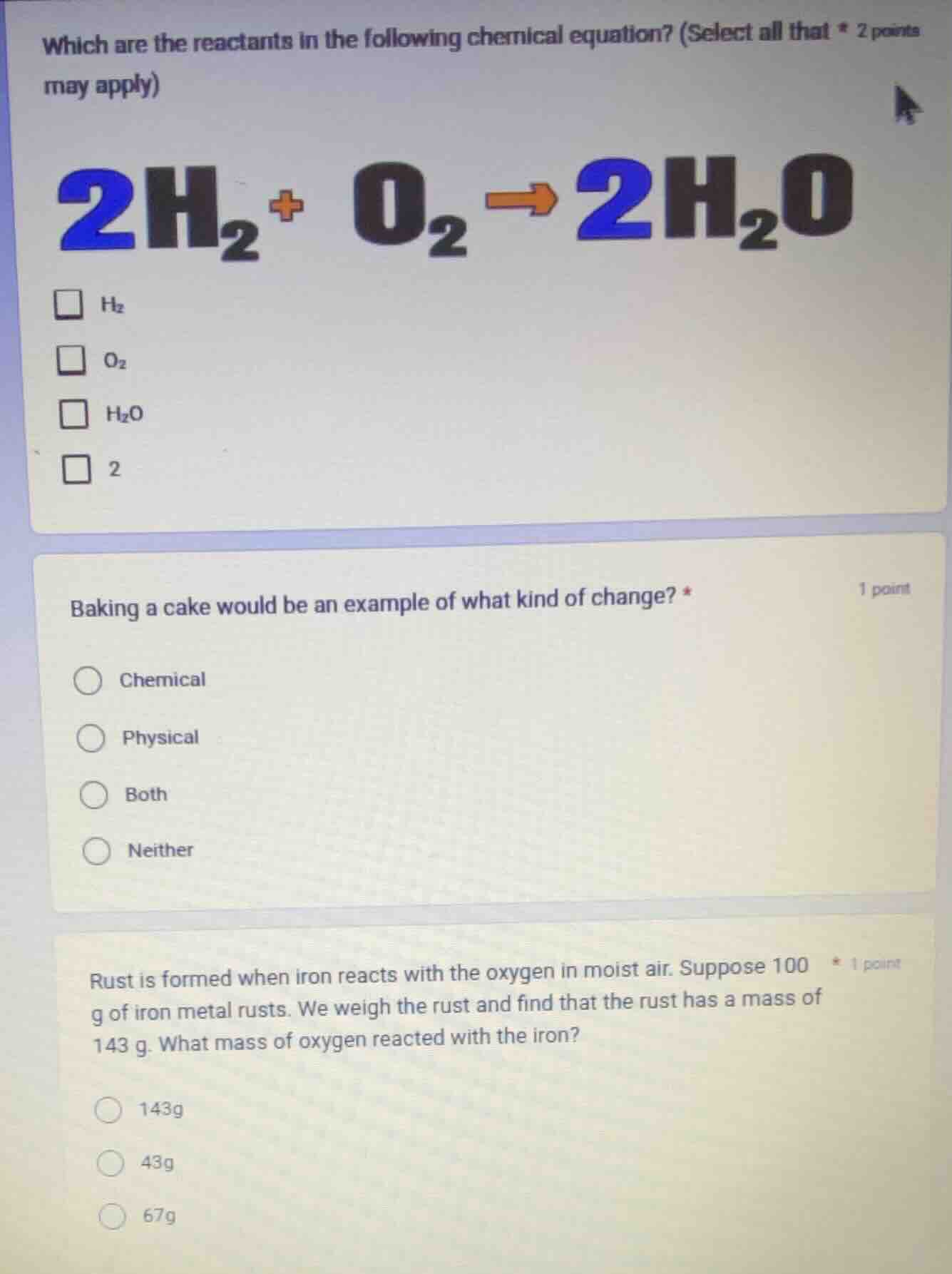
which are the reactants in the following chemical equation? (select all that may apply) * 2 points
$ce{2h_{2} + o_{2} -> 2h_{2}o}$
$square$ $ce{h_{2}}$
$square$ $ce{o_{2}}$
$square$ $ce{h_{2}o}$
$square$ 2
baking a cake would be an example of what kind of change? * 1 point
$\bigcirc$ chemical
$\bigcirc$ physical
$\bigcirc$ both
$\bigcirc$ neither
rust is formed when iron reacts with the oxygen in moist air. suppose 100 g of iron metal rusts. we weigh the rust and find that the rust has a mass of 143 g. what mass of oxygen reacted with the iron? * 1 point
$\bigcirc$ 143g
$\bigcirc$ 43g
$\bigcirc$ 67g
First Question
In a chemical equation, reactants are on the left side of the arrow, the substances that undergo reaction. The coefficient (the number 2) is not a reactant, and $\text{H}_2\text{O}$ is a product on the right side.
Baking a cake involves irreversible chemical reactions (like protein denaturation, Maillard reactions) that create new substances, so it is a chemical change.
Step1: Apply Law of Conservation of Mass
The mass of products equals total mass of reactants. Let $m_{\text{O}_2}$ = mass of oxygen.
$\text{Mass of rust} = m_{\text{Fe}} + m_{\text{O}_2}$
Step2: Solve for oxygen mass
Rearrange to isolate $m_{\text{O}_2}$.
$m_{\text{O}_2} = \text{Mass of rust} - m_{\text{Fe}}$
$m_{\text{O}_2} = 143\ \text{g} - 100\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{H}_2$, $\text{O}_2$
---