QUESTION IMAGE
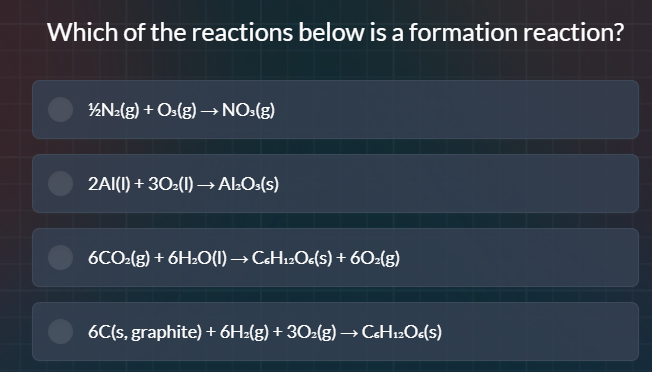
Question
which of the reactions below is a formation reaction?
½n₂(g) + o₃(g) → no₃(g)
2al(l) + 3o₂(l) → al₂o₃(s)
6co₂(g) + 6h₂o(l) → c₆h₁₂o₆(s) + 6o₂(g)
6c(s, graphite) + 6h₂(g) + 3o₂(g) → c₆h₁₂o₆(s)
To determine a formation reaction, we recall that a formation reaction is one where 1 mole of a compound is formed from its constituent elements in their standard states (most stable form at 1 atm and 25°C). Let's analyze each option:
Option 1: $\boldsymbol{\frac{1}{2}N_2(g) + O_3(g)
ightarrow NO_3(g)}$
- Ozone ($O_3$) is not the standard state of oxygen. The standard state of oxygen is $O_2(g)$. So this is not a formation reaction.
Option 2: $\boldsymbol{2Al(l) + 3O_2(l)
ightarrow Al_2O_3(s)}$
- The standard state of aluminum is solid ($Al(s)$), not liquid ($Al(l)$). Also, the standard state of oxygen is $O_2(g)$, not $O_2(l)$. So this is not a formation reaction.
Option 3: $\boldsymbol{6CO_2(g) + 6H_2O(l)
ightarrow C_6H_{12}O_6(s) + 6O_2(g)}$
- This is the reverse of a combustion reaction (or photosynthesis), and it involves reactants that are compounds ($CO_2$ and $H_2O$), not elements. Formation reactions require elements as reactants, so this is not a formation reaction.
Option 4: $\boldsymbol{6C(s, \text{graphite}) + 6H_2(g) + 3O_2(g)
ightarrow C_6H_{12}O_6(s)}$
- Carbon is in its standard state (graphite, $C(s, \text{graphite})$), hydrogen is in its standard state ($H_2(g)$), and oxygen is in its standard state ($O_2(g)$). These are the constituent elements of glucose ($C_6H_{12}O_6$), and 1 mole of glucose is formed. This matches the definition of a formation reaction.
A formation reaction forms 1 mole of a compound from its elements in standard states. The fourth reaction uses C (graphite), H₂(g), and O₂(g) (standard states) to form 1 mole of C₆H₁₂O₆(s), while others fail (non - standard states or compound reactants).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
6C(s, graphite) + 6H₂(g) + 3O₂(g) → C₆H₁₂O₆(s) (the fourth option)