QUESTION IMAGE
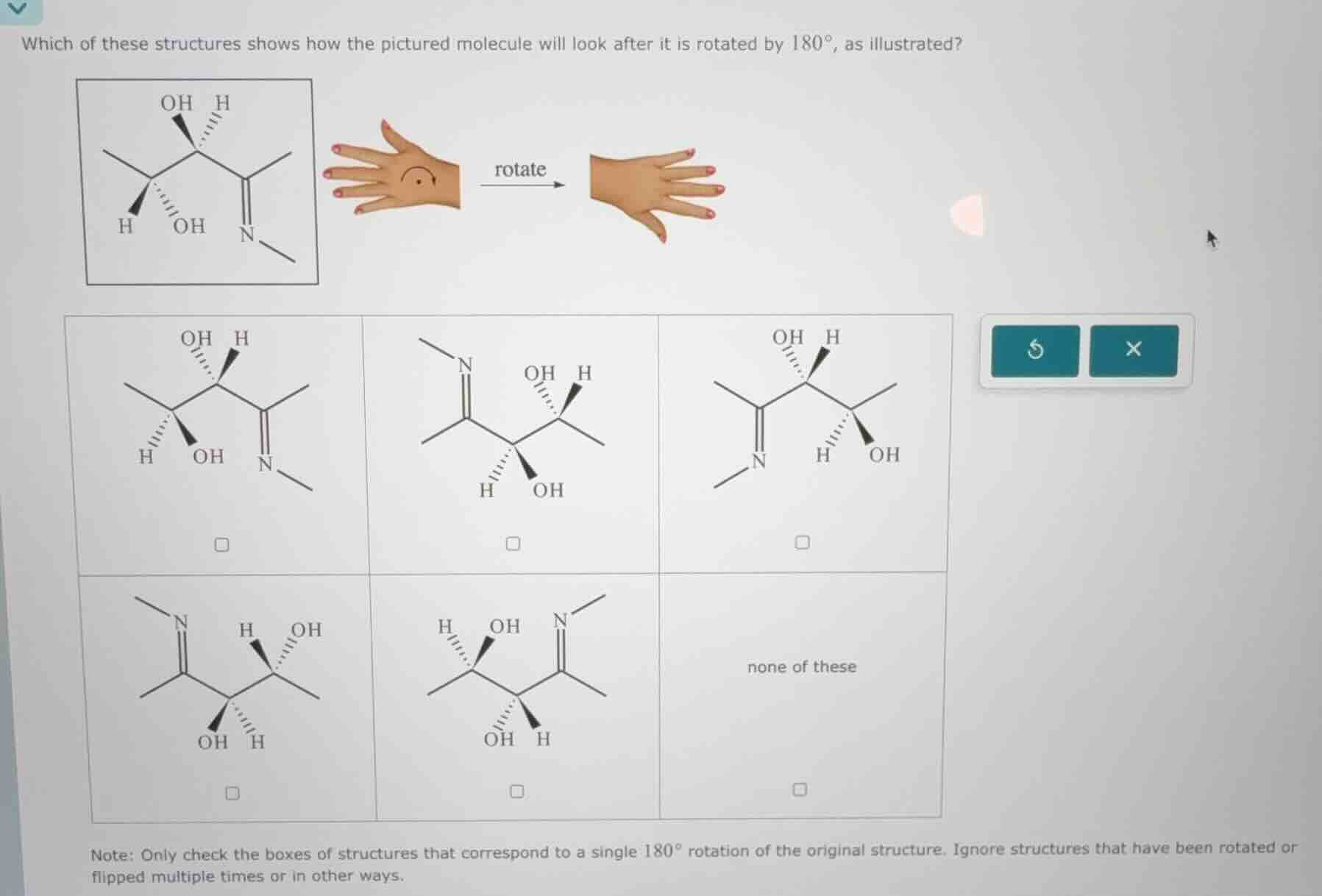
Question
which of these structures shows how the pictured molecule will look after it is rotated by $180^{\circ}$, as illustrated?note: only check the boxes of structures that correspond to a single $180^{\circ}$ rotation of the original structure. ignore structures that have been rotated or flipped multiple times or in other ways.
Step1: Identify rotation effect
A 180° rotation inverts the position of all substituents relative to the chiral centers, and reverses the direction of wedged/dashed bonds for each group while maintaining the overall connectivity of the molecule.
Step2: Match original to rotated structure
Compare the original molecule:
- Left chiral center: $CH_3$ (wedged), $H$ (dashed), $OH$ (wedged), $CH(OH)C(CH_3)=NCH_3$
- Right chiral center: $C(CH_3)=NCH_3$, $H$ (wedged), $OH$ (dashed), $CH(CH_3)OH$
After 180° rotation, the rightmost structure in the top row (third option) matches: the $C(CH_3)=NCH_3$ group moves to the left end, and all wedged/dashed bonds are inverted appropriately to reflect the 180° turn, while the connectivity of atoms remains identical to the original.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Third top option:
OH H
\ /
C(CH3)=N-C-C(CH3)
\ /
H OH
\
CH3(Structural description: Left end is $C(CH_3)=NCH_3$, followed by chiral center with wedged $H$, dashed $OH$, then chiral center with wedged $OH$, dashed $H$, ending with $CH_3$)