QUESTION IMAGE
Question
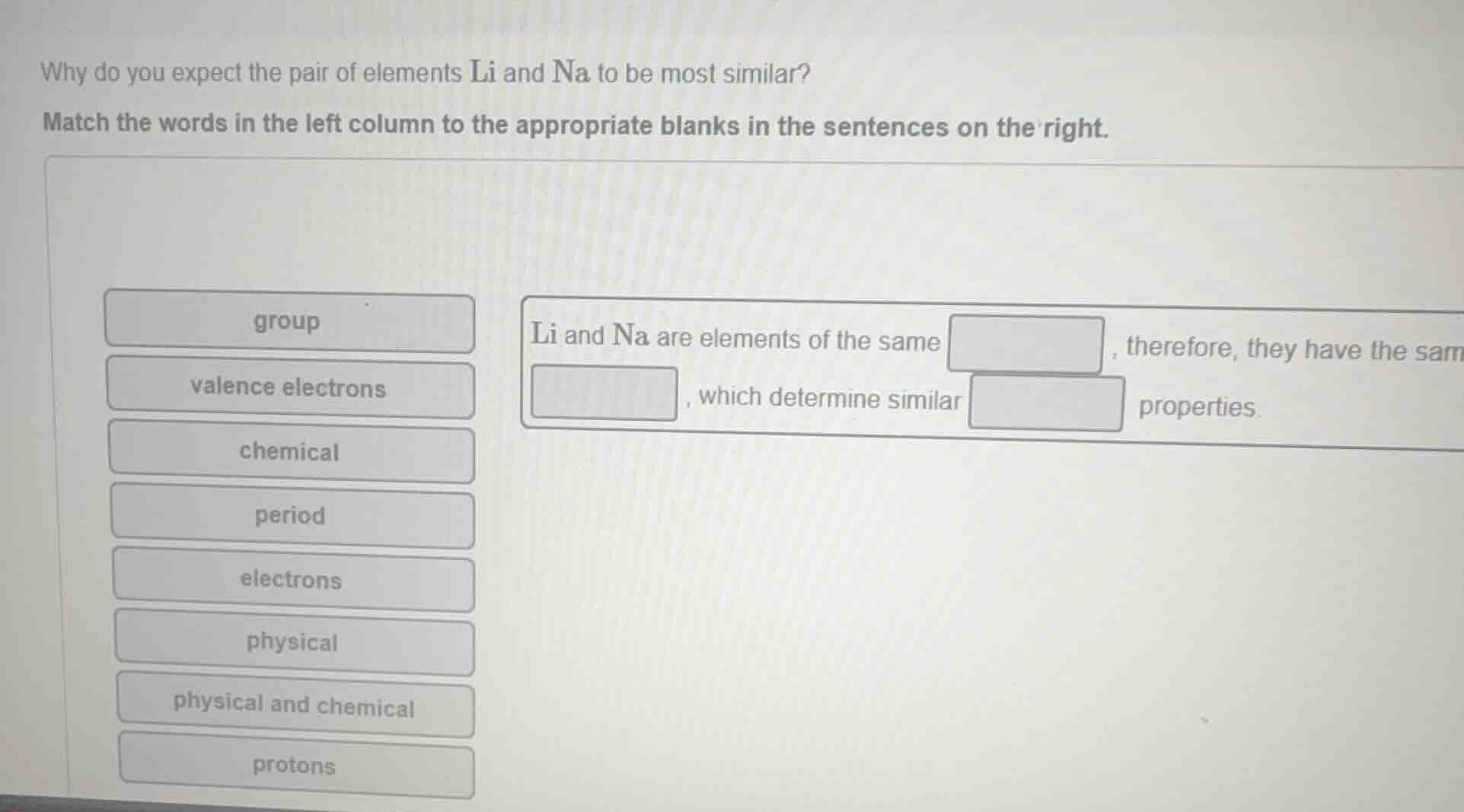
why do you expect the pair of elements li and na to be most similar? match the words in the left column to the appropriate blanks in the sentences on the right. group li and na are elements of the same , therefore, they have the sam valence electrons , which determine similar properties. chemical period electrons physical physical and chemical protons
Lithium (Li) and Sodium (Na) are both in Group 1 of the periodic table. Elements in the same group have the same number of valence electrons, which are the electrons responsible for an element's chemical behavior, leading to similar chemical properties.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Li and Na are elements of the same group, therefore, they have the same valence electrons, which determine similar chemical properties.