QUESTION IMAGE
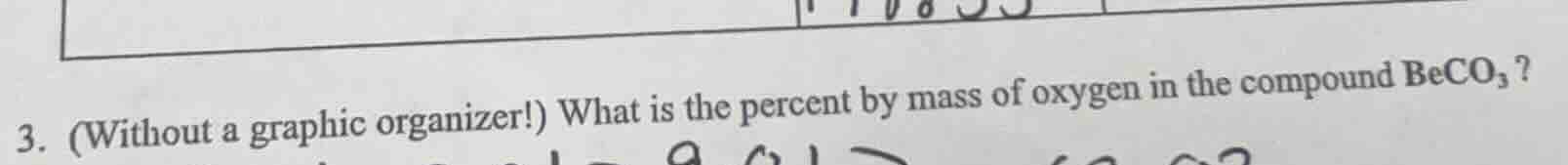
Question
- (without a graphic organizer!) what is the percent by mass of oxygen in the compound $\ce{beco_{3}}$?
Step1: Calculate molar mass of $\text{BeCO}_3$
First, find atomic masses: $\text{Be}=9.01\ \text{g/mol}$, $\text{C}=12.01\ \text{g/mol}$, $\text{O}=16.00\ \text{g/mol}$.
$$\begin{align*}
M(\text{BeCO}_3)&=9.01 + 12.01 + 3\times16.00\\
&=9.01 + 12.01 + 48.00\\
&=69.02\ \text{g/mol}
\end{align*}$$
Step2: Find total mass of oxygen
$$
m(\text{O})=3\times16.00=48.00\ \text{g/mol}
$$
Step3: Compute mass percent of oxygen
Use formula: $\text{Mass Percent}=\frac{\text{Total Mass of Element}}{\text{Molar Mass of Compound}}\times100\%$
$$
\text{Mass Percent of O}=\frac{48.00}{69.02}\times100\%\approx69.5\%
$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Approximately 69.5%