QUESTION IMAGE
Question
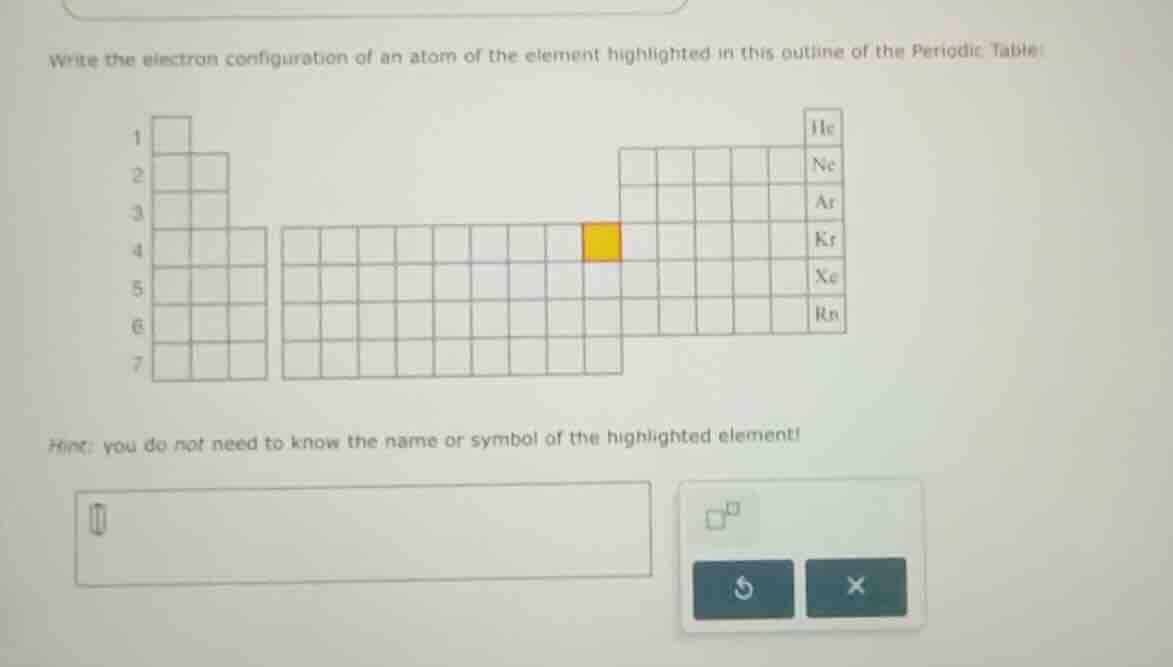
write the electron configuration of an atom of the element highlighted in this outline of the periodic table
hint: you do not need to know the name or symbol of the highlighted element!
Step1: Identify the period and group
The highlighted element is in period 4 (row 4) and in the d - block (transition metals) and also considering the noble gas core. The noble gas before period 4 is Ar (argon, electron configuration $1s^2 2s^2 2p^6 3s^2 3p^6$). Then, for period 4, the element is in the d - block. The number of electrons in the 4s and 3d orbitals: first, 4s is filled with 2 electrons, and then the 3d orbitals. Looking at the position, the element is in the 4th period, and the d - block. The general electron configuration for a transition metal in period 4, with Ar as the noble gas core, is $[Ar] 4s^2 3d^x$. Now, to find x: the position in the d - block. The d - block has 10 elements (from 3d¹ to 3d¹⁰). The highlighted element is the 8th element in the d - block of period 4? Wait, no, let's count the columns. The first two columns are s - block (group 1 and 2), then the d - block starts. The noble gas Ar is at the end of period 3. Then period 4: 4s², then 3d. The highlighted element: let's count the columns from the start of the d - block. The d - block in period 4: the first d - block element is Sc (3d¹), then Ti (3d²), V (3d³), Cr (3d⁵, 4s¹), Mn (3d⁵, 4s²), Fe (3d⁶), Co (3d⁷), Ni (3d⁸), Cu (3d¹⁰, 4s¹), Zn (3d¹⁰, 4s²). Wait, maybe a better way: the electron configuration of the element. The noble gas core is Ar ($[Ar]$), then 4s², and then 3d. Now, the position of the element: in the periodic table, the row is period 4, and the column in the d - block. Let's see the periodic table outline: the noble gases are He (1), Ne (2), Ar (3), Kr (4), Xe (5), Rn (6). The highlighted element is in period 4, to the left of Kr. The s - block (period 4) is 4s², then d - block. The number of electrons in 3d: let's count the number of columns from the start of the d - block. The d - block has 10 columns. The element is in the 8th column of the d - block? Wait, no, maybe I made a mistake. Wait, the correct way: the electron configuration for an element in period 4, d - block. The noble gas before period 4 is Ar. So the electron configuration is $[Ar] 4s^2 3d^6$? Wait, no, let's think again. Wait, the element is in period 4, and the position: let's count the number of electrons. Ar has 18 electrons. Then, 4s² (2 electrons, total 20), then 3d. The highlighted element: let's see the periodic table structure. The first two columns (group 1, 2) are s - block (4s¹, 4s²), then the next 10 columns are d - block (3d¹ to 3d¹⁰), then p - block. The highlighted element is in the d - block, period 4. Let's count the number of d - electrons. The element is the 8th element in the d - block? Wait, no, let's look at the standard periodic table. The element with electron configuration $[Ar] 4s^2 3d^6$ is Fe (iron), but maybe the highlighted element is Fe? Wait, no, let's check the position. Alternatively, maybe the element is in the 4th period, and the electron configuration is $[Ar] 4s^2 3d^6$. Wait, but let's do it step by step.
- Determine the noble gas core: The noble gas preceding period 4 is Ar (argon), with electron configuration $1s^2 2s^2 2p^6 3s^2 3p^6$.
- Determine the valence electrons: For period 4, the element is in the d - block. The 4s orbital is filled before the 3d orbital (Aufbau principle). So 4s² is filled first. Then, the 3d orbitals. The number of 3d electrons: the element is in the 8th position of the d - block (counting from the start of the d - block in period 4). Wait, the d - block has 10 elements (3d¹ to 3d¹⁰). If we count the columns, the first d - block element (Sc) is 3d¹, Ti (3d²), V (3d³), Cr (3d⁵, 4s¹), Mn (3d⁵,…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{[Ar] 4s^2 3d^8}$ (or if the element is Fe, it would be $[Ar] 4s^2 3d^6$, but based on the position in the periodic table outline, the correct electron configuration is $[Ar] 4s^2 3d^8$ for Ni - like position)