QUESTION IMAGE
Question
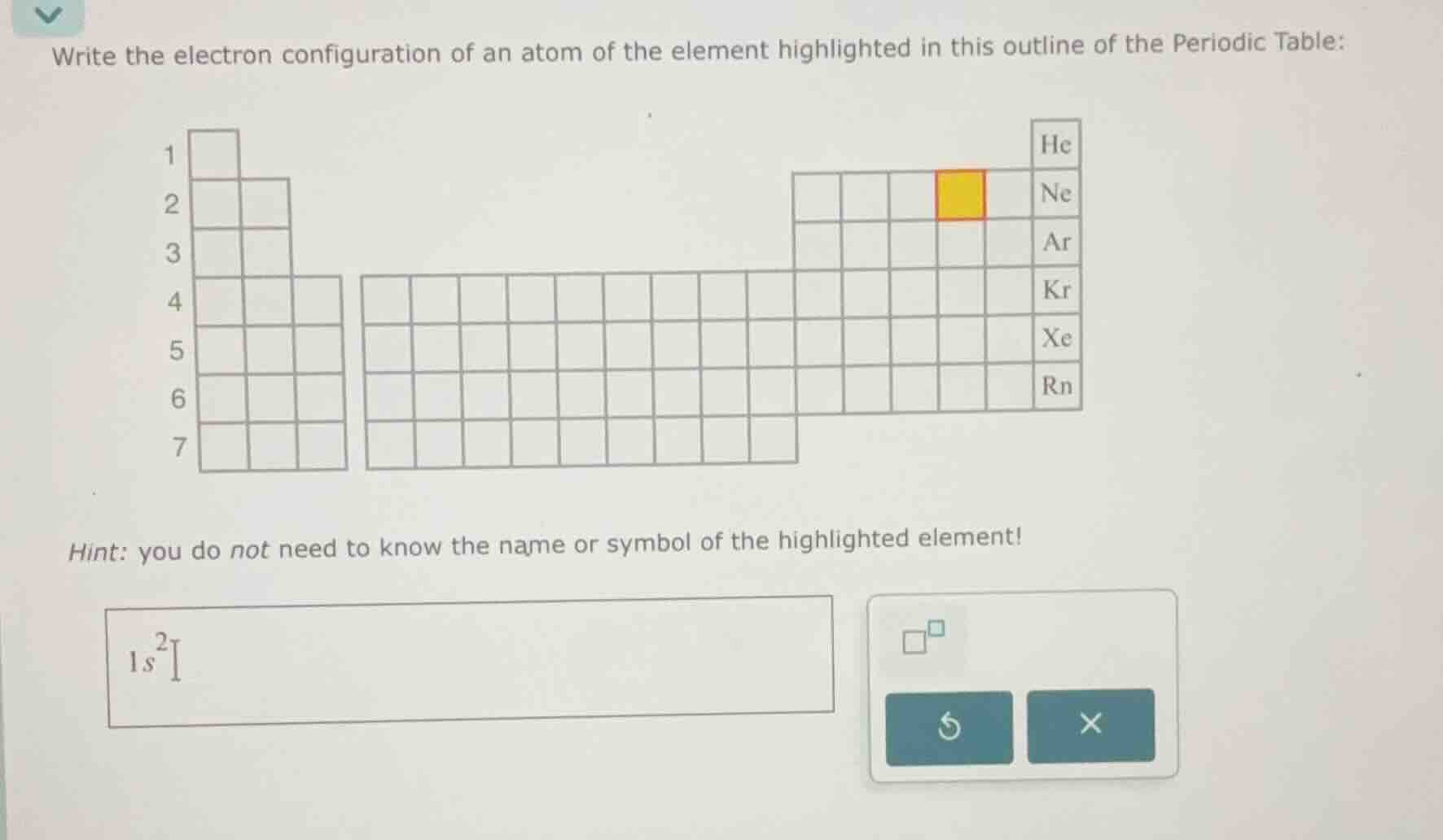
write the electron configuration of an atom of the element highlighted in this outline of the periodic table:
hint: you do not need to know the name or symbol of the highlighted element!
$1s^{2}$
Step1: Identify element's period/group
The highlighted element is in Period 2, Group 16 (16th column, counting from left: Group 1, 2, then 13-18; this is the 4th element in the p-block of Period 2). Total electrons = atomic number = 8.
Step2: Fill orbitals by Aufbau principle
Fill 1s first: $1s^2$. Then 2s: $2s^2$. Remaining electrons: $8 - 2 - 2 = 4$, fill 2p: $2p^4$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$1s^22s^22p^4$